What are the colors of the three most intense lines in the emission spectrum of hydrogen
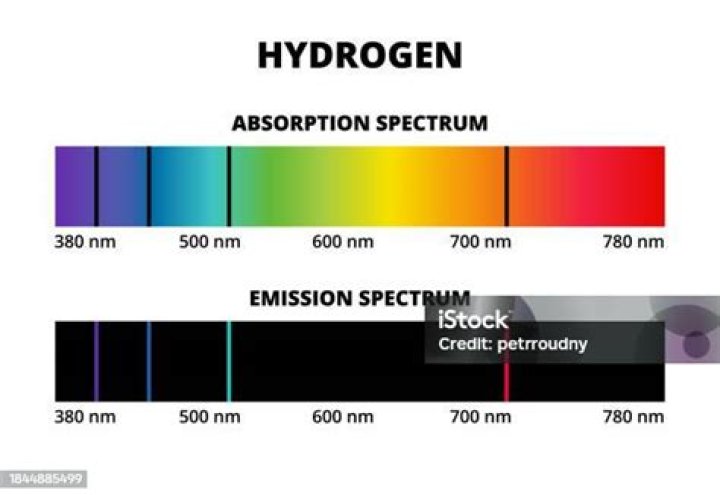
He noted that hydrogen gas does not emit a continuous spectrum, but rather discrete lines. The three most prominent lines are violet, blue-green, and red.
What 3 Colours will be observed in the emission spectrum of hydrogen?
WavelengthColor656.2red486.1blue-green434.0blue-violet410.1violet
Which color line from the hydrogen spectrum has the highest energy?
Violet waves have the most energy of the visible spectrum.
What colors are the four most intense lines in the emission spectrum of hydrogen?
(b) When the light emitted by a sample of excited hydrogen atoms is split into its component wavelengths by a prism, four characteristic violet, blue, green, and red emission lines can be observed, the most intense of which is at 656 nm.Which color emission line has the highest energy?
When it comes to visible light, the highest frequency color, which is violet, also has the most energy. The lowest frequency of visible light, which is red, has the least energy.
Why is there no yellow line in the hydrogen emission spectrum?
Each time an electron changes from a higher energy state to a lower one, a package of energy is emitted. Sometimes the energy emitted is visible. There is no transition in the hydrogen atom that results in the emission of yellow light.
What are the colors produced by hydrogen?
- Yes, hydrogen is an invisible gas. …
- Green hydrogen, blue hydrogen, brown hydrogen and even yellow hydrogen, turquoise hydrogen and pink hydrogen. …
- Depending on the type of production used, different colours are assigned to the hydrogen.
How do electrons create the colors in a line emission spectrum?
How do electrons create the colors in a line-emission spectrum? The colors are created by photons containing specific amounts of energy, released when an electron makes the transition from a higher energy level to a lower energy level. … Energy level, orbital shape, orientation of the orbital and spin.What do the different colors of the line spectrum represent?
Because each element has a different set of emission colors from the emission spectrum. … They represent the wavelengths of light that is absorbed by the spectrum They represent the energy that the atom gives off when their electrons are in place. They also represent frequency and energy.
What color is emitted by N 4 to N 2?Transition of n3→24→2NameH-α / Ba-αH-β / Ba-βWavelength (nm, air)656.279486.135Energy difference (eV)1.892.55ColorRedAqua
Article first time published onWhich color spectral line in the hydrogen line spectrum has the lowest energy?
The visible photons in the hydrogen spectrum are the Balmer series lines. The lowest energy and longest wavelength photon corresponds to the 3→2 transition and is red.
Which Colour has the highest wavelength?
The colour Red has the longest wavelength and violet has the shortest wavelength in the visible spectrum.
Which color in the visible spectrum has the highest frequency?
Violet waves have the highest frequencies.
Which light is highest energy?
Gamma rays have the highest energies, the shortest wavelengths, and the highest frequencies.
What is the Colour of h2 gas?
Hydrogen is a colourless gas – depending on the production type, different colours are assigned to the hydrogen. Green hydrogen is produced by electrolysis of water, using only electricity from renewable energies.
What is blue and green hydrogen?
Green hydrogen is produced using electrolysis of water, and blue hydrogen utilizes natural gas. Green hydrogen represents a major opportunity for governments and private business to harness a valuable, sustainable energy resource in the coming decades.
What color is liquid hydrogen?
Hydrogen is colorless as a liquid. Its vapors are colorless, odorless, tasteless, and highly flammable.
How many lines does hydrogen emission spectrum have?
So, there are four lines in the visible spectrum of hydrogen. There are, of course, lots of other lines in hydrogen’s spectrum but they are all outside of our visible range so we cannot see them with our naked eye (and a diffraction grating).
Why does hydrogen only emit 4 colors?
Although hydrogen has only one electron, it contains many energy levels. When its electron jumps from higher energy level to a lower one, it releases a photon. Those photons cause different colours of light of different wavelengths due to the different levels. Those photons appear as lines.
How many spectral lines does hydrogen have?
The electron energy level diagram for the hydrogen atom. He found that the four visible spectral lines corresponded to transitions from higher energy levels down to the second energy level (n = 2).
What do the different Coloured lines in a line spectrum represent Why are the spectra for each element unique?
When atoms are excited they emit light of certain wavelengths which correspond to different colors. The emitted light can be observed as a series of colored lines with dark spaces in between; this series of colored lines is called a line or atomic spectra. Each element produces a unique set of spectral lines.
Which are the three spectral lines in the Balmer series hydrogen used in astronomy?
Balmer lines are historically referred to as “H-alpha”, “H-beta”, “H-gamma” and so on, where H is the element hydrogen.
Why are there different colors emitted?
When you heat an atom, some of its electrons are “excited* to higher energy levels. When an electron drops from one level to a lower energy level, it emits a quantum of energy. … The different mix of energy differences for each atom produces different colours.
What specific color of light has the longest wavelength How about the shortest wavelength?
WAVELENGTHS OF VISIBLE LIGHT As the full spectrum of visible light travels through a prism, the wavelengths separate into the colors of the rainbow because each color is a different wavelength. Violet has the shortest wavelength, at around 380 nanometers, and red has the longest wavelength, at around 700 nanometers.
Which color of light does the smallest energy drop of an electron produce?
Red has the smallest energy drop because it drops from the 3rd energy level to the first.
What color light is emitted when the electron drops from n 3 to n 2?
The difference between these two energy levels will then give us the actual energy of the red-light photon that is emitted when an electron drops from the n = 3 level to the n = 2 level.
What is the wavelength of light emitted from the n 3 to n 2?
Electron TransitionEnergy (J)Wavelength (nm)Balmer Series (to n=2)n=3 to n=23.03 x 10-19656n=4 to n=24.09 x 10-19486n=5 to n=24.58 x 10-19434
What color is emitted by n 6 to n 2?
n=6 to n=2, violet light, 410 nm. b. Explain why this transition involves the most energy based on your understanding of the attractive forces between the electrons and protons in the atom. As the electron moves toward the nucleus of the atom there is a stronger attraction.
Which color of the below colors of spectral lines would be the highest energy?
Red is the lowest energy visible light and violet is the highest. A solid object has color depending on the light it reflects. If it absorbs light in the red and yellow region of the spectrum, it will have a blue color. Here is an example.
Which color in the visible spectrum of hydrogen has the highest frequency the lowest frequency?
When it comes to visible light, the highest frequency color, which is violet, also has the most energy. The lowest frequency of visible light, which is red, has the least energy.
Which of the wavelengths most likely corresponds to the wavelength of the most intense line in Xenon's emission spectrum?
the wavelength that corresponds to the most intense line lies around 500-520 nm.