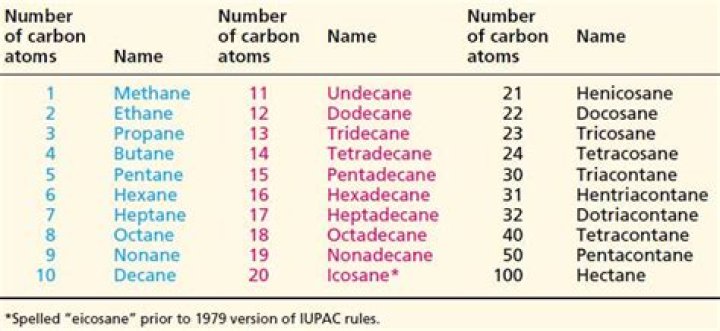
What are the first 50 alkanes
Methane (CH4)Ethane (C2H6)Propane (C3H8)Butane (C4H10)Pentane (C5H12)Hexane (C6H14)Heptane (C7H16)Octane (C8H18)
What are the names of the first 10 alkanes in order?
- Methane.
- Ethane.
- Propane.
- Butane.
- Pentane.
- Hexane.
- Heptane.
- Octane.
What are examples of alkanes?
Methane (CH4), ethane (C2H6), propane (C3H8) and butane (C4H10) are the first four alkanes. Methane gas, whose molecular formula is CH4, is the simplest alkane.
What are the 7 alkanes?
Number of C atomsNumber of isomersName of straight chain53n-pentane65n-hexane79n-heptane818n-octaneWhat are the first 10 alkynes?
NameMolecular FormulaPropyneC3H41-ButyneC4H61-PentyneC5H81-HexyneC6H10
What are the first 8 hydrocarbons?
methaneCH4pentaneC5H12hexaneC6H14heptaneC7H16octaneC8H18
What is the longest alkane?
The longest linear alkane is now C59H120, which is considerably longer than expected from the force-field results and remarkably different to the PM3 result.
What is an 11 carbon alkane called?
undecane (11 carbons) dodecane (12 carbons)How do you remember the first 10 alkanes?
For example the first 10 alkanes in order are , Methane, Ethane, Propane, Butane, Pentane, Hexane, Heptane, Octane, Nonane and Decane. These can be memorised with “Many elephants prefer blue pinapples.
What are the 12 alkanes?NameMolecular Formula (CnH2n + 2)Number of Possible IsomerspropaneC3H8—butaneC4H102pentaneC5H123hexaneC6H145
Article first time published onWhat is the name of an alkane with 22 carbon atoms?
Table 1. Summary of the Straight-Chain AlkanesNameFormulaNumber of Structural IsomersNonaneC9H2036DecaneC10H2275UndecaneC11H24
Which is the simplest alkane?
The simplest alkane is methane. Methane contains only one carbon atom and four hydrogen atoms.
Is petrol an alkane?
Gasoline is a mixture of alkanes from pentane up to about decane. Kerosene contains alkanes from about n=10 to n=16. … Alkanes with higher values of n are found in diesel fuel, fuel oil, petroleum jelly, paraffin wax, motor oils, and for the highest values of n, asphalt.
Are all hydrocarbons alkanes?
NamebutaneMolecular Formula (CnH2n + 2)C4H10Condensed Structural FormulaCH3CH2CH2CH3Number of Possible Isomers2
Why alkane is called paraffin?
Alkanes have only single bonds which are sigma covalent. … Hence, alkanes are called paraffins because they have lesser affinity towards general reagents. In other words they are inert, not readily active.
What are the first 5 alkenes?
- Ethene (C2H4)
- Propene (C3H6)
- Butene (C4H8)
- Pentene (C5H10)
- Hexene (C6H12)
- Heptene (C7H14)
- Octene (C8H16)
- Nonene (C9H18)
What are the 10 prefixes in chemistry?
NumberPrefix7Hepta-8Octa-9Nona-10Deca-
How many alkanes are there?
AlkaneNonaneMolecular FormulaC9H20CompositionCH3-7(CH2)-CH3Carbon atoms in chain9Hydrogen atoms in chain20
What is a 12 carbon chain called?
Alkanes – saturated hydrocarbons. The names of the straight chain saturated hydrocarbons for up to a 12 carbon chain are shown below. The names of the substituents formed by the removal of one hydrogen from the end of the chain is obtained by changing the suffix -ane to -yl.
What is a branched alkane?
A branched chain alkane or branched alkane is an alkane which has alkyl groups bonded to its central carbon chain. Branched alkanes contain only carbon and hydrogen (C and H) atoms, with carbons connected to other carbons by single bonds only, but the molecules contain branches (methyl, ethyl, etc.)
How many isomers do alkanes have?
Structural Or Constitutional Isomers Structural Isomers are molecules which have the same molecular formula but have different connectivities (The Order They Are Put Together). Alkanes can be very simple examples of this. With the structural formula C4H10 there are two different isomers possible.
What are the first alkanes?
The first four alkanes are methane (CH4), ethane (C2H6), propane (C3H8) and butane (C4H10). The simplest alkane is the gas methane, whose molecular formula is CH4.
What are the first 4 alkenes?
NameNumber of Carbon atomsMolecular Formula CnH2nethene2C2H2(2) = C2H4Propene3C3H2(3) = C3H6Butene4C4H2(4) = C4H8Pentene5C5H2(5) = C5H10
What are the first ten organic prefixes?
PrefixNumber of Carbon atomsFormulameth-1Ceth-2C2prop-3C3but-4C4
Is ch3 a methyl?
A methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms — CH3. In formulas, the group is often abbreviated Me. … It is a very stable group in most molecules.
How do you remember the first 4 alkanes?
The first four alkanes are: methane, ethane, propane and butane. A mnemonic to help you remember the order of the alkanes: mice eat paper bags.
What is the name of the alkane having five carbons?
Alkanes with five or more carbon atoms are named by adding the suffix -ane to the appropriate numerical multiplier, except the terminal -a is removed from the basic numerical term. Hence, C5H12 is called pentane, C6H14 is called hexane, C7H16 is called heptane and so forth.
How many hydrogens does an alkane with 17 carbons have?
An allane with 17 carbons has 36 hydrogens.
How are carbons numbered?
Carbon atoms are numbered beginning from the reactive end of the molecule, the CHO (aldehyde) or “C” double bonded “O” (carbonyl) end of the molecule. Each carbon atom is then numbered in order through the end of the chain. … We then number the remaining carbons in order through the end of the chain.
What is the name of this alkane CH3 CH3?
Ethane is an alkane comprising of two carbon atoms.
What is alkyl group?
Definition: An alkyl is a functional group of an organic chemical that contains only carbon and hydrogen atoms, which are arranged in a chain. Examples include methyl CH3 (derived from methane) and butyl C2H5 (derived from butane).