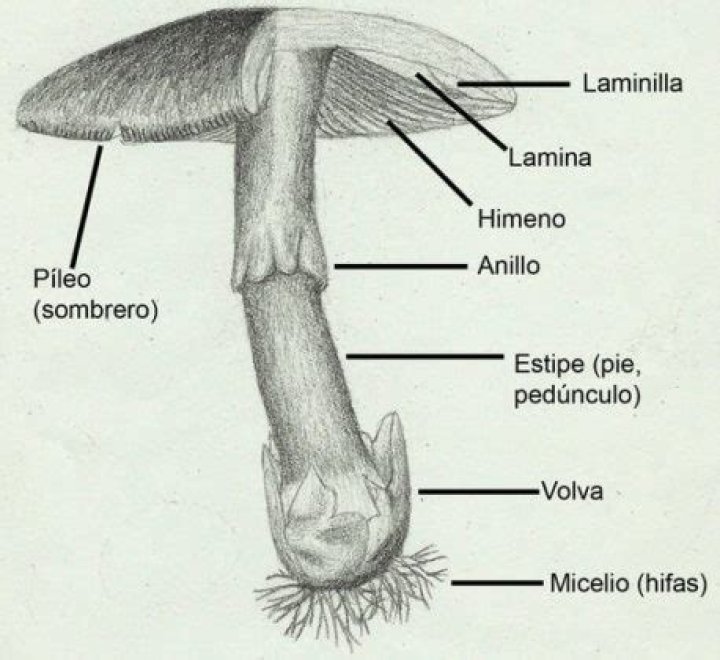
What are the IR range of OH group
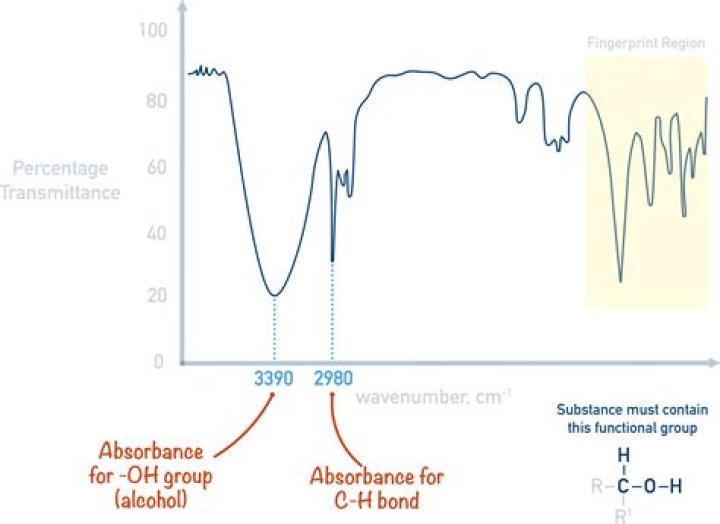
Absorption (cm-1)AppearanceGroup3300-2500strong, broadO-H stretching3200-2700weak, broadO-H stretching3000-2800strong, broadN-H stretching3333-3267strong, sharpC-H stretching
Why are oh peaks broad in IR?
The reason that the O–H stretch band of carboxylic acids is so broad is becase carboxylic acids usually exist as hydrogen-bonded dimers. One of the most distinct and easily recognizable peaks in an IR spectrum is the broad O-H absorption of alcohols and phenols.
What are the peaks in IR spectrum?
An IR -spectrum routinely shows peaks from the range of 3600 to 500 cm-1. IR -frequencies correspond to the frequencies of molecular vibrations. Molecules vibrate at many frequencies, because each vibration involves a particular group of atoms, and a particular motion such as stretches, bending, wagging etc.
What does OH stretch mean?
The O-H stretch is usually a broad IR band at 3400-3200 cm-1. This is the case if you run the IR spectrum of the pure liquid alcohol where the concentration is high. Carbonyl compounds sometimes give rise to a weak C=O. … Hydrogen-bonded O-H stretches are much broader because the hydrogen bonds vary in strength.What does IR peak at 3000 mean?
Absorption peaks above 3000 cm-1 are frequently diagnostic of unsaturation. Alkynyl C-H Stretch.
Why are NH or OH peaks broad?
The main challenge is differentiating between the OH Peaks and that of the NH peaks since they mostly overlap and appear at various deltas depending on the functional group whether amides, arenes, alcohols, phenols, aryl amides and co so with the two the main difference is the fact that the OH Peaks are mostly broader …
What is a strong peak in IR?
A strong peak around 1450 cm-1 indicates the presence of methylene groups (CH2), while an additional strong peak about 1375 cm-1 is caused by a methyl group (CH3) (examples 1, 8-10).
Why are OH and NH stretches broad?
The reason is again extent of intermolecular hydrogen bonding, which is greater due to higher electronic density on the O. But apart from that, the general reason for broadening the spectra is the extent of intermolecular hydrogen bonding, which is somewhat lesser for normal amines.Why large number of peaks generates in IR spectrum?
Hydrogen bonding between hydroxyl groups leads to some variations in O-H bond strength, which results in a range of vibrational energies. The variation results in the broad peaks observed.
What is the OH stretching frequency of phenol?In the solid phase (Nujol mull) phenols, which are not intramolecular, exhibit intermolecular hydrogen-bonded hydroxyl groups, v(OH···OH), in the range 3180–3400cm-1. The 2-alkylphenols exhibit v(OH···OH), in the range 3438–3535 cm-1, and the frequency increases with the increasing steric factor of the alkyl group.
Article first time published onHow will you distinguish ketone and amide functional groups by using IR absorption?
IR spectroscopy readily identifies the carbonyl group C=O. of organic compounds: of amides, or esters, or ketones, of acids as a strong sharp absorption at around 1900−1700 cm−1 . … Sometimes you can infer an ester if you see a carbonyl, and note strong absorption at approx. 1050 cm−1 due to the C−O stretch.
What is the typical wavenumber range of a carbonyl group in IR?
Carbonyl stretching peaks generally fall between 1900 and 1600 cm-1 (assume all peak positions hereafter are in wavenumber units), a relatively unique part of the IR spectrum. This area is sometimes referred to as the carbonyl stretching region as a result.
How many peaks in an IR spectrum would you expect to see that would characterize an alcohol functional group?
characterize an alcohol functional group? There is a total of 2 peaks that will be expected.
What is a broad IR peak?
A very broad peak in the region between 3100 and 3600 cm-1 indicates the presence of exchangeable protons, typically from alcohol, amine, amide or carboxylic acid groups (see further discussion of this below).
Why do tertiary amines have no peak in the 3200 3600 cm-1 region?
IR Spectroscopy Tutorial: Amines Secondary amines (R2NH) show only a single weak band in the 3300-3000 cm-1 region, since they have only one N–H bond. Tertiary amines (R3N) do not show any band in this region since they do not have an N–H bond.
What does an IR spectrum show?
The IR spectra tell you what types of vibrational modes (motion) the molecule responds with after it absorbs that light, and when you figure out which peaks correspond to which motions, you can figure out what functional groups the molecule has and (almost) what the molecule is.
Which compound would you expect to show an intense IR absorption at 3300 cm-1?
CH3CH2CONH2 would be expected to show the observed peaks.
What does high transmittance mean in IR?
Infrared frequencies correspond to vibrational modes in specific chemical bonds. … High transmittance at a frequency means there are few bonds to absorb that “color” light in the sample, low transmittance means there is a high population of bonds which have vibrational energies corresponding to the incident light.
Why are NH peaks broad in NMR?
Broad peaks can represent inhomogeneities in the magnetic field which may have been caused by poor shimming, paramagnetic materials in the sample or particulate matter. Alternatively, peaks can broaden due to exchange processes on the NMR time scale.
Do OH groups show up NMR?
Unlike most other types of protons, protons on oxygen and nitrogen are quite variable in where they show up in the 1H NMR spectrum. The –OH signal of the same compound can be at a different chemical shift if the concentration of the sample changes. … Thus as concentration changes, so does chemical shift.
Do alcohol hydrogens show up on NMR?
All alcohols, such as ethanol, are very, very slightly acidic. The hydrogen on the -OH group transfers to one of the lone pairs on the oxygen of the water molecule. … Deuterium atoms don’t produce peaks in the same region of an NMR spectrum as ordinary hydrogen atoms, and so the peak disappears.
What affects intensity of IR peaks?
The most important factor that influences the intensity of an IR absorption band is the change in dipole moment that occurs during a vibration. For example, an aldehyde C=O. stretch usually occurs near 1730 cm⁻¹. An alkene C=C stretch usually occurs near 1650 cm⁻¹.
Why is in IR spectrum of benzoyl chloride a weak band near 1750 cm-1 is formed?
Why in the IR spectrum of Benzoyl chloride, a weak band near 1750 cm-1 is formed? A weak band near 1750 cm-1. Since, band is of weak intensity, it must be due to fermi Resonance. So correct option is Fermi resonance between C = O band and first overtone.
Does OH or NH have higher stretching frequency?
The Nature of Vibrational Spectroscopy Consequently, C-H, N-H and O-H bonds have much higher stretching frequencies than do corresponding bonds to heavier atoms. … Thus, the stretching frequency of a free O-H bond is 3600 cm-1, but the O-D equivalent is lowered to 2600 cm-1.
What are the broad regions in an IR spectrum called?
The infrared portion of the electromagnetic spectrum is usually divided into three regions; the near-, mid- and far- infrared, named for their relation to the visible spectrum.
What is the effect of hydrogen bonding on the position and shape of an O-H stretch?
Cooperative hydrogen bonding increases the O-H bond length whilst causing a 20-fold greater reduction in the H····O and O····O distances [436]. The increase in bond length has been correlated with the hydrogen bond strength and resultant O-H stretch vibrations [1318].
Where does a carbonyl C O stretch appear in an IR spectrum Mcq?
When using IR spectroscopy, carbonyl (C=O) groups display characteristic peaks at approximately 1700cm-1, while alcohol groups (O-H) display characteristic peaks around 3300cm-1.
Where is aromatic ring on IR?
Aromatic hydrocarbons show absorptions in the regions 1600-1585 cm-1 and 1500-1400 cm-1 due to carbon-carbon stretching vibrations in the aromatic ring.
Which of the following range represents IR finger print region?
The region of the infrared spectrum from 1200 to 700 cm-1 is called the fingerprint region.
What is the absorbance of an IR peak with a 25 transmittance?
TransmittanceAbsorbance (AU)35%0.45630%0.52325%0.60220%0.699
What does OH stretch mean?
The O-H stretch is usually a broad IR band at 3400-3200 cm-1. This is the case if you run the IR spectrum of the pure liquid alcohol where the concentration is high. Carbonyl compounds sometimes give rise to a weak C=O. … Hydrogen-bonded O-H stretches are much broader because the hydrogen bonds vary in strength.