What bonds are found in water
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. The attraction between individual water molecules creates a bond known as a hydrogen bond.
How many covalent bonds are present in water?
Each pair of shared electrons represents one covalent bond, so two covalent bonds hold the water molecule together.
Why does ice float on water?
Believe it or not, ice is actually about 9% less dense than water. Since the water is heavier, it displaces the lighter ice, causing the ice to float to the top.
What are the bond angles in water?
The actual bond angle in the water molecule is 104.5°.How many covalent bonds are there in 9g of H2O?
of covalent bonds in 9 grams of water . One molecule of water has 2 covalent bonds so you have to find the no. of molecules in 9 grams of water and then multiply it with 2 🙂 ***** follow me *****
How many bonds does oxygen have?
12. Oxygen atoms form 2 covalent bonds because oxygen atoms have 6 valence electrons (2 lone pairs plus 2 unpaired electrons that are shared to achieve octet).
How many bonds are there in ammonia?
In NH3 molecule, three covalent bonds are present. Hydrogen atom has 1 valence electron. Nitrogen atom has 5 valence electrons.
Can you sink in the Dead Sea?
You’ll notice the buoyancy as soon as you get into the water. You only have to be a foot or so deep and you can sit down, lean back, and float. It is actually impossible to sink or swim in the Dead Sea.Is H2O bent?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom. …
Why is water polar?Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Article first time published onIs ice lighter than liquid water?
When water freezes, water molecules form a crystalline structure maintained by hydrogen bonding. Solid water, or ice, is less dense than liquid water. Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart, which lowers the density.
How many molecules of water are there in 12g of ice?
H20 has a molar mass of 18.02g/mol. Use this and the mass to find the number of moles. Your answer should be: 4.01×10^23 molecules!
What is the total number of atoms present in 9g of water?
Grade 12. 9 is 1/2 of 18, meaning that 1/2 mole is present in 9 g of water. 1/2 * 6.022 *10^23 = 3.011 * 10^23 molecules H2O. However, if you want to know how many ATOMS are present in 3.011 *10^23, you must then multiply by 3 (2 hydrogen atoms and 1 oxygen atom per molecule), and get 9.033 * 10^23 atoms.
How do you find the total number of covalent bonds?
Atom (Group number)Number of BondsNumber of Lone PairsFluorine (Group 17 or 7A)13
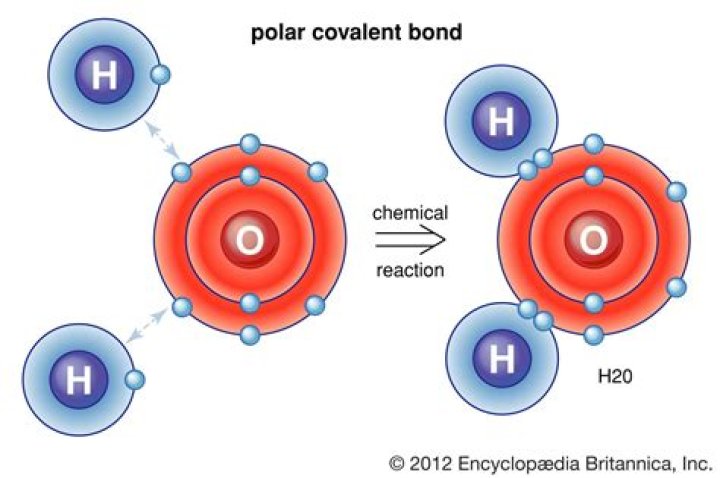
Is water covalent bond?
A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
Is h2o a double bond?
Now, in water, i.e. H2O, oxygen is the central atom and can either form one double or two single bonds. We have two hydrogens which can only form single bonds. Thus, in water, oxygen is singly bonded to two hydrogen atoms.
Is CO2 a double bond?
The CO2 molecule has 2 double bonds so minus 2 electrons from the final total. So the overall total number of electrons should be 2, this is the electron region number.
How many bonds does chlorine have?
AtomValenceBromine1Chlorine1Iodine1Oxygen2
Can you have 3 bonds?
A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. … The most common triple bond, that between two carbon atoms, can be found in alkynes. Other functional groups containing a triple bond are cyanides and isocyanides.
How many bonds can p form?
The reason that phosphorus can form “five bonds” and nitrogen only three or four has to do with the size of the two atoms.
What is the angle between atoms in water?
A water molecule consists of two hydrogen atoms and one oxygen atom. The three atoms make an angle; the H-O-H angle is approximately 104.5 degrees.
What is the shape of water chemistry?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
How many bonding and lone pairs are in H2O?
AB2E2: Water, H2O A water molecule consists of two bonding pairs and two lone pairs (see Figure 10).
How many charge clouds are there in H2O?
Charge Clouds# of BondsExample42H2O55PF54SF43ClF3
How many electron groups are in H2O?
H2O: Water has four electron groups so it falls under tetrahedral for the electron-group geometry. The four electron groups are the 2 single bonds to Hydrogen and the 2 lone pairs of Oxygen. Since water has two lone pairs it’s molecular shape is bent.
Why is h20 not linear?
In water, the oxygen atom has two lone pairs. These two lone pairs repel the hydrogen-oxygen bonded pairs so much that the molecule is at its lowest energy arrangement when the H-O-H bond angle is 104.5 degrees. As a result, the water molecule can be classified as non-linear.
What is sticky water?
The extraordinary stickiness of water is due to the two hydrogen atoms, which are arranged on one side of the molecule and are attracted to the oxygen atoms of other nearby water molecules in a state known as “hydrogen bonding.” (If the molecules of a liquid did not attract one another, then the constant thermal …
What molecules make up water?
A water molecule has three atoms: two hydrogen (H) atoms and one oxygen (O) atom.
Can you float forever?
“The trick is to not panic; as long as you don’t panic, you can float forever, until you are rescued or until you find the energy to swim to shore.”
Which sea you Cannot drown?
The Dead Sea is landlocked and in the lowest valley on earth. All the minerals of the surrounding countryside get washed into one pool, which in turn gets baked by the sun. This concentrates the salts so much so that the Dead Sea becomes times as salty as the ocean, which defines why people don’t drown in the dead sea.
In which lake we can't sink?
Dead SeaLocationWestern AsiaCoordinates31°30′N 35°30′ECoordinates: 31°30′N 35°30′ELake typeEndorheic HypersalineNative name[ים המלח (in Hebrew) البحر الميت (in Arabic)] Error: {{Native name}}: missing language tag (help) (language?)



