What determines IR absorption

In general, IR absorption is caused by the interaction between the IR electric field vector and the molecular dipole transition moments related to the molecular vibrations. Absorption is at a maximum when the electric field vector and the dipole transition moment are parallel to each other.
What is measured in IR spectroscopy?
IR spectroscopy is the measurement of the wavelength and intensity of the absorption of infrared light by a sample (Putzig et al., 1994).
What factors determine IR frequency?
The amount of energy required to stretch a bond depends on the strength of the bond and the masses of the bonded atoms. The stronger the bond, the greater the energy required to stretch it. The frequency of the vibration is inversely proportional to the mass of the atoms, so heavier atoms vibrate at lower frequencies.
What is absorbance in IR?
Absorbance: Units used to measure the amount of IR radiation absorbed by a sample. Absorbance is commonly used as the Y axis units in IR spectra. Absorbance is defined by Beer’s Law, and is linearly proportional to concentration.Can gases be measured by IR spectrometry?
In infrared (IR) spectroscopy, radiation in the IR region of the electromagnetic spectrum is passed through a gas sample. … It cannot detect diatomic or noble gases such as O2, N2, H2, He, or Ar, which do not have absorbance bands in the infrared region of the spectrum.
How does infrared spectroscopy determine purity?
“They” would periodically take an infrared spectrum of the sample and compare it with a spectrum of the pure ester. Any extraneous peaks would be caused by the presence of impurities. If the peaks matched those of known impurities, they could use the intensities to calculate the purity of the ester.
Which detector is used in IR spectroscopy?
The lead sulphide detector is used for the near-infrared region of the spectrum. For mid- and far-infrared radiation the mercury cadmium telluride detector is used.
What is the absorbance of an IR peak with a 50% transmittance?
TransmittanceAbsorbance (AU)60%0.22255%0.2650%0.30145%0.347How do you measure absorbance?
Absorbance is measured using a spectrophotometer or microplate reader, which is an instrument that shines light of a specified wavelength through a sample and measures the amount of light that the sample absorbs.
What is infrared wavelength?Wavelength range and sources Infrared radiation (IR), also known as thermal radiation, is that band in the electromagnetic radiation spectrum with wavelengths above red visible light between 780 nm and 1 mm. IR is categorized as IR-A (780 nm-1.4 µm), IR-B (1.4-3 µm) and IR-C, also known as far-IR (3 µm-1 mm).
Article first time published onWhat is a strong IR peak?
That peak a little after 1700 cm-1 is the C=O. stretch. When it’s present, the C=O. stretch is almost always the strongest peak in the IR spectrum and impossible to miss.
What affects IR absorption intensity?
The absorption bands in IR spectra have different intensity, that can usually be referred to as strong (s), medium (m), weak (w), broad and sharp. The intensity of a absorption band depends on the polarity of the bond, the bond with higher polarity will show more intense absorption band.
Why are IR peaks broad?
The sharpness or broadness of a stretch in IR spectra depends on extent of Hydrogen bonding present in the molecule. Basically, if it undergoes immense intermolecular hydrogen bonding, the peaks tend to be broader and the lesser the hydrogen bonding becomes, the sharper the peaks get in the spectra.
Which bonds shown strongest absorption in the IR?
The C=O bond of simple ketones, aldehydes, and carboxylic acids absorb around 1710 cm-1. Usually, it’s the strongest IR signal. Carboxylic acids will have O-H also.
What is difference between IR and FTIR?
FTIR spectrometers have several prominent advantages: (1) The signal-to-noise ratio of spectrum is significantly higher than the previous generation infrared spectrometers. (2) The accuracy of wavenumber is high. … Due to these advantages, FTIR Spectrometers have replaced dispersive IR spectrometers.
Which of the following molecules Cannot be Analysed by IR spectroscopy?
While the generation of heat is a probable event following the absorption of infrared radiation, it is important to distinguish between the two. Infrared is a form of radiation that can travel through a vacuum while heat is associated with the motion and kinetic energy of molecules.
What is FTIR gas analyzer?
A FTIR gas analyser collects a full infrared spectrum continuously, allowing for hundreds of gases to be detected and measured at once. Changes are only needed to software calibration files to measure new gases.
What are the types of IR detectors?
The two main types of detectors are thermal and photonic (photodetectors). The thermal effects of the incident IR radiation can be followed through many temperature dependent phenomena. Bolometers and microbolometers are based on changes in resistance. Thermocouples and thermopiles use the thermoelectric effect.
Why are IR detectors cooled?
They allow cooling down the detector with low efficiency and not to the very low temperature. They do not generate any vibrations and therefore are suitable for thermal detectors, where the microphone effect can decrease their performance. Photon detectors need to be cooled down even to 77K or better.
What is the Mull technique?
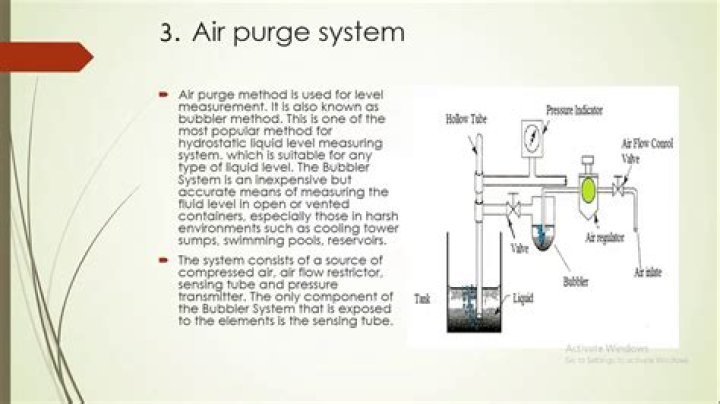
a) Mull technique: In this technique, the finely crushed sample is mixed with Nujol (mulling agent) in n a marble or agate mortar, with a pestle to make a thick paste. A thin film is applied onto the salt plates. This is then mounted in a path of IR beam and the spectrum is recorded.
Can IR detect impurities?
Infrared spectroscopy is used in research to identify samples, do quantitative analysis, or detect impurities. Infrared spectroscopy can be used on gaseous, liquid, or solid samples and does not destroy the sample in the process.
How do you determine the purity of an ester?
You can check the purity of your ester by distilling the product and collecting the distillate at the boiling point of the ester. Record the yield again.
How do you determine the purity of an organic compound?
The classical criteria for determining the purity of organic compounds are correct elemental compositions (Section 1-1A) and sharpness of melting point or constancy of boiling point. Important though these analytical and physical criteria are, they can be misleading or even useless.
How does spectrometer measure absorbance?
Spectrophotometry is a method to measure how much a substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. The basic principle is that each compound absorbs or transmits light over a certain range of wavelength.
What is the unit of absorbance?
Absorbance is measured in absorbance units (Au), which relate to transmittance as seen in figure 1. For example, ~1.0Au is equal to 10% transmittance, ~2.0Au is equal to 1% transmittance, and so on in a logarithmic trend.
How is UV absorption measured?
A UV/VIS spectrophotometer measures the intensity of light passing through a sample solution in a cuvette, and compares it to the intensity of the light before it passes through the sample.
How is transmittance measured?
To calculate transmittance, a measurement of the source (incident flux, fi) is acquired by placing it in line with the detector without the sample in place. The sample then is placed between the source and detector, and the transmitted light, ft, is measured.
How do you calculate transmittance?
Transmittance (T) is the fraction of incident light which is transmitted. In other words, it’s the amount of light that “successfully” passes through the substance and comes out the other side. It is defined as T = I/Io, where I = transmitted light (“output”) and Io = incident light (“input”).
What is the lambda max?
Lambda max (λmax): The wavelength at which a substance has its strongest photon absorption (highest point along the spectrum’s y-axis). This ultraviolet-visible spectrum for lycopene has λmax = 471 nm.
What is the frequency range for infrared?
Infrared radiation (IR radiation for short) describes electromagnetic waves in the spectral range between visible red light and longer-wave microwave radiation (also known as terahertz radiation). Infrared (IR) has wavelengths λ between 780 nm and 1 mm, which corresponds to a frequency range from 300 GHz to 400 THz.
What is the range of UV?
The UV region covers the wavelength range 100-400 nm and is divided into three bands: UVA (315-400 nm) UVB (280-315 nm) UVC (100-280 nm).