What does furfural taste like
Furfural (FEMA# 2489, CAS# 98-01-1) is the most significant flavor compound generated during the heating of sugars in nature. The profile of furfural is, unsurprisingly, quite brown and caramelic. Underneath the obvious caramel character lurks more than a hint of the much harsher bitter almond note.
Why is furfural toxic?
Furfural is a skin, mucous membrane, and respiratory irritant. The main toxicity targets are respiratory system, nervous system, liver, and kidneys. Furfural is not classifiable regarding its carcinogenicity to humans. Furfural is moderately toxic to aquatic species.
What is the use of furfural?
Furfural is used as a selective solvent for refining lubricating oils and rosin, and to improve the characteristics of diesel fuel and catalytic cracker recycle stocks.
Is furfural natural?
Furfural is a natural dehydration product of xylose, a pentose sugar often found in large quantities in the hemicellulose fraction of lignocellulosic biomass. This heterocyclic and aromatic aldehyde consists of a furan ring with an aldehyde side group.Is furfural an alcohol?
NamesOdorburning odorDensity1.128 g/cm3Melting point−29 °C (−20 °F; 244 K)Boiling point170 °C (338 °F; 443 K)
How is furfural made?
Furfural is produced by removing water from or dehydrating five-carbon sugars such as xylose and arabinose. These pentose sugars are commonly obtained from the hemicellulose fraction of biomass wastes like cornstalks, corncobs and the husks of peanuts and oats.
Is furfural volatile?
NamesVapor pressure2 mmHg (20 °C)Magnetic susceptibility (χ)−47.1×10−6 cm3/molHazardsFlash point62 °C (144 °F; 335 K)
How do you make a furfural solution?
- Patent number: 9181211.
- Abstract: Furfural is produced by mixing an aqueous feedstock solution containing C5 sugar and/or C6 sugar with a heated high boiling, water-miscible solvent, such as sulfolane, and a solid acid catalyst. …
- Type: Grant.
- Filed: December 28, 2012.
What is the main safety concern when working with furfural?
* Exposure to high concentrations can cause you to become dizzy, lightheaded, and to pass out. * Furfural may cause a skin allergy. If allergy develops, very low future exposure can cause itching and a skin rash.
Is furfural biodegradable?CSTR systems in which furfural was continuously added resulted in 80% of the theoretically expected biogas. Intermediates in the anaerobic biodegradation of furfural were determined by spike additions in serum-bottle assays using the enriched consortium from the CSTR systems.
Article first time published onWho discovered furfural?
This is a natural process that is the basis of the commercial production of furfural. In the first half of the 19th century, chemists such as Johann W. Döbereiner at the University of Jena (Germany) and John Stenhouse at Glasgow University discovered furfural in the distillation products of various biomaterials.
What is the Colour of furfural?
Freshly distilled furfural is colorless, but when it is exposed to the atmosphere for some time, it turns via yellow and brown to black.
What foods contain furfuryl alcohol?
Furfuryl alcohol has been found in a wide variety of products, including wheat, bran, coffee, seafood, nuts, processed produce, legumes, alcohol and honey.
Which heteroatom is present furfural?
NamesBeilstein Reference103221ChEBICHEBI:35559ChEMBLChEMBL278980ChemSpider7738
Is furfural a functional group?
Furfural is an aldehyde of furan and is a yellow oily liquid in pure form, but tends to turn brown upon prolonged exposure to air and moisture. An aldehyde is an organic functional group that has a carbonyl group (carbon-oxygen double bond) attached to a hydrogen and some other carbon-based side chain.
What is meant by furfural extraction?
The Furfural Refining process is a solvent extraction process used to remove undesirable components of low lubricating oil quality naturally present in crude oil distillate and residual stocks. The unit produces paraffinic or naphthenic raffinates suitable for further processing into lube base stocks.
What does furfuryl alcohol smell like?
Furfuryl Alcohol is a colorless or yellow liquid that turns red or brown in air. It has a mild Alcohol or Ether-like odor and is used as a flavoring and in making sealants and cements. * Furfuryl Alcohol is on the Hazardous Substance List because it is regulated by OSHA and cited by ACGIH, DOT, NIOSH and NFPA.
Why is furfuryl alcohol in food?
Furfuryl alcohol is a naturally occurring byproduct of heating and appears in many foods and beverages. Furfuryl alcohol forms as a result of baking that gives our buns the characteristic brown color tops. It is not added to our products.
Is furfural soluble in water?
Furthermore, furfural itself is also used as a solvent in some reactions due to its physical–chemical characteristics. It was excellently described by Eseyin and Steele as follows: “Furfural is commonly used as a solvent; it is soluble in ethanol and ether and somewhat soluble in water.
What is the time weighted average exposure limit for furfural?
The mean time-weighted average personal exposure to furfural was 0.13 mg/m3 (25 observations) and ranged from undetectable to 0.23 mg/m3.
Is Methanoic acid Formic acid?
formic acid (HCO2H), also called methanoic acid, the simplest of the carboxylic acids, used in processing textiles and leather.
What is the Colour of the furfural derivative formed with glucose?
Furfuraldehyde and the pentoses, arabinose and xylose, give only an intense yellow color with this reagent. The disaccharides give the colors of the simple sugars of which they are composed; thus maltose and lactose give the amethyst color of glucose, inulin gives the blue of levulose, and sucrose gives a mixed color.
Which product is formed when furfural react with ag2o?
Furfural was oxidized to furoic acid by molecular oxygen under catalysis by 150 nm-sized Ag2O/CuO (92%) or simply CuO (86.6%).
Which hydro atom is present in furfural?
Furfural is an aldehyde that is furan with the hydrogen at position 2 substituted by a formyl group.
Is a furan aromatic?
Furan and Furan-Related Compounds. Furan is an aromatic compound with the participation of the oxygen lone pair in the π-electron system to satisfy Hückel’s rule, 4n + 2 (n = 1) electrons. The compound is stable to heating up to about 550°C (depending also on heating time).
Does styrene react with water?
Styrene is primarily a synthetic chemical. It is also known as vinylbenzene, ethenylbenzene, cinnamene, or phenylethylene. … It often contains other chemicals that give it a sharp, unpleasant smell. It dissolves in some liquids but doesn’t dissolve easily in water.
When a mixture of furan and ammonia is passed over heated alumina at 753K What is obtained?
A None of above. When a mixture of Furan and ammonia is passed over heated alumina at 753K. a) Pyrrole is obtained.
What is HMF in honey?
HMF (HydroxyMethylFurfuraldehyde) is used as an indicator of heat and storage changes in honey. HMF is formed by the breakdown of fructose in the presence of an acid. Heat increases the speed of this reaction. … HMF occurs naturally in most honeys and usually increases with the age and heat treatment of honey.
What are the precursors for furfural production?
Furfural was not formed from HMF or l-ascorbic acid in vinegar. Instead, ribose, xylose, arabinose, galacturonic acid, glucuronic acid, and pentosan were the main precursors.
How do you make furfuryl alcohol?
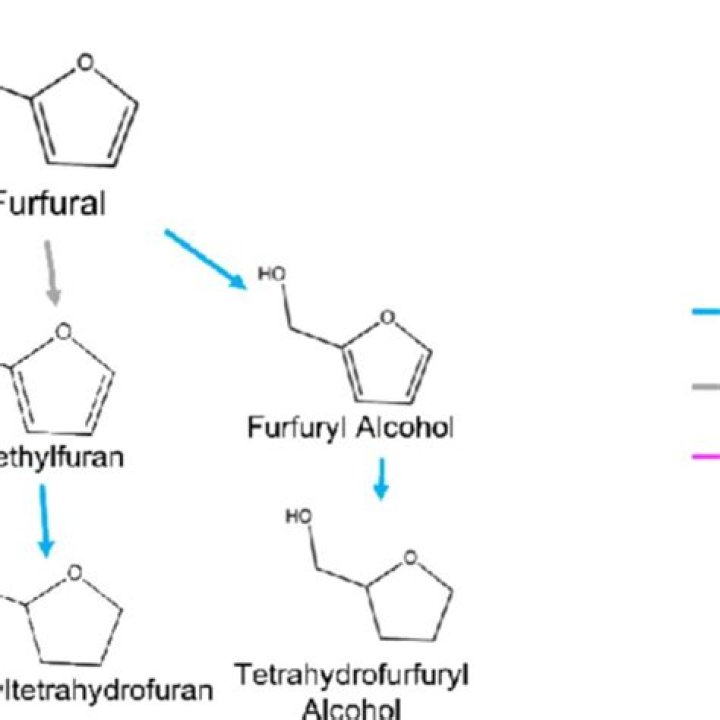
Furfuryl alcohol can be produced by hydrogenation of furfural (FAL) in the gas phase over a copper chromite catalyst. Furfural itself can be produced sustainably from agricultural wastes such as corn cobs, bagasse, etc.
What are carcinogens?
A carcinogen is an agent with the capacity to cause cancer in humans. Carcinogens may be natural, such as aflatoxin, which is produced by a fungus and sometimes found on stored grains, or manmade, such as asbestos or tobacco smoke. Carcinogens work by interacting with a cell’s DNA and inducing genetic mutations.



