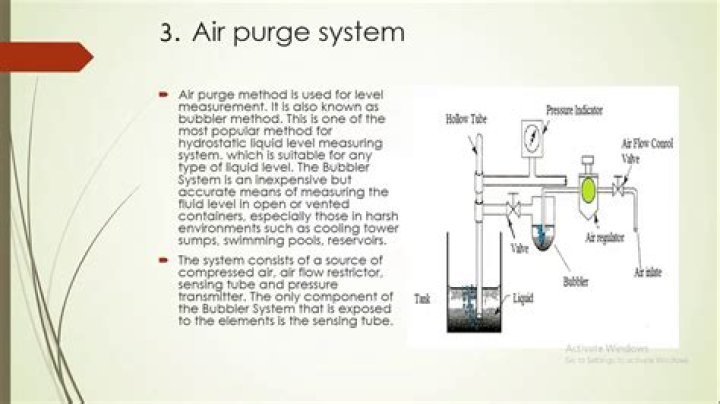
What element has a mass of 50
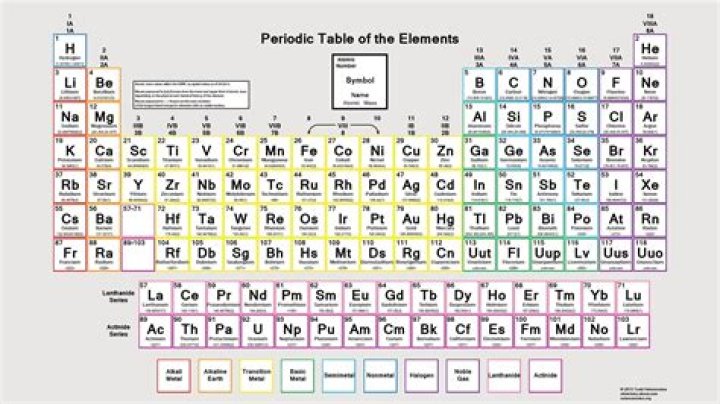
Atomic MassName chemical elementnumber112.411Cadmium48114.818Indium49118.71Tin50121.76Antimony51
What element has 24 protons and 52 mass number?
Chromium 52: The atomic number Z = 24, so there are 24 protons and 24 electrons. The mass number A = 52.
What is the mass of CR 52?
Compound Formula52CrBoiling Point2672 °CDensity7.14 g/cm3Solubility in H2ON/AExact Mass51.940507472
What element has a mass of 54?
Iron, isotope of mass 54.What is the atomic notation of chromium 52?
Properties of Chromium-52 Isotope:CHROMIUM-52Relative Isotopic Mass51.9405075Neutron Number (N)28Atomic Number (Z)24Mass Number (A)52
What is the mass number of zinc 68?
Properties of Zinc-68 Isotope:ZINC-68Relative Isotopic Mass67.9248442Neutron Number (N)38Atomic Number (Z)30Mass Number (A)68
What is the mass of CR 54?
PubChem CID11579113Molecular FormulaCrSynonyms14304-97-3 Chromium-54 54Cr Chromium, isotope of mass 54 (~54~Cr)Chromium More…Molecular Weight53.938877DatesModify 2021-12-05 Create 2006-10-26
Is Chromium-52 stable?
Chromium has four stable isotopes, 50, 52, 53, and 54, and isotope values are typically reported in delta notation as a ratio of 53Cr to 52Cr, which are the most abundant Cr isotopes. Chromium is highly redox-sensitive, existing as the mobile Cr(VI)O42 − (chromate) anion in oxygenated systems.What is the mass number of zinc 67?
Zinc-67 is the stable isotope of zinc with relative atomic mass 66.927131, 4.10 atom percent natural abundance and nuclear spin 5/2.
Is Chromium 51 man made?Chromium-51 is a synthetic radioactive isotope of chromium having a half-life of 27.7 days and decaying by electron capture with emission of gamma rays (0.32 MeV); it is used to label red blood cells for measurement of mass or volume, survival time, and sequestration studies, for the diagnosis of gastrointestinal …
Article first time published onWhat is the mass of he4?
Helium-4 atom is the stable isotope of helium with relative atomic mass 4.002603.
What is number 50 on the periodic table?
Tin is a chemical element with the symbol Sn (from Latin: stannum) and atomic number 50.
What type of element is element 86?
Radon is element 86 in the periodic table and is one of the noble gases along with helium, neon, argon, krypton and xenon. Radon exists in a number of isotopic forms and its most stable form is radon-222. Radon is colourless, odourless and inert which means that it does not react with materials or other gases.
What element has a mass of 222?
PubChem CID25000466Molecular FormulaRaSynonymsRadium, isotope of mass 222 13967-62-9 Radium-222 DTXSID50109552Molecular Weight222.01537DatesModify 2021-12-18 Create 2008-10-27
Is RB 85 stable?
Rubidium 85 Chloride is a stable (non-radioactive) isotope of Rubidium. It is both naturally occurring and produced by fission.
Is Copper 62 stable?
Natural copper comprises two stable isotopes, 63Cu and 65Cu, and 5 principal radioisotopes for molecular imaging applications (60Cu, 61Cu, 62Cu, and 64Cu) and in vivo targeted radiation therapy (64Cu and 67Cu).
How many neutrons does iron 56 have?
GeneralProtons26Neutrons30Nuclide dataNatural abundance91.754%
What is the defined mass of a proton?
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
What isotope has 30 protons?
Explanation: Each zinc isotope contains 30 protons, 30 massive, positively charged nuclear particles. That Z=30 , (Z=the atomic number) , defines the nucleus as ZINC.
How do u find the mass of an element?
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
How is atomic weight calculated?
The atomic weight of any atom can be found by multiplying the abundance of an isotope of an element by the atomic mass of the element and then adding the results together. This equation can be used with elements with two or more isotopes: Carbon-12: 0.9889 x 12.0000 = 11.8668.