What hybridization is bromine
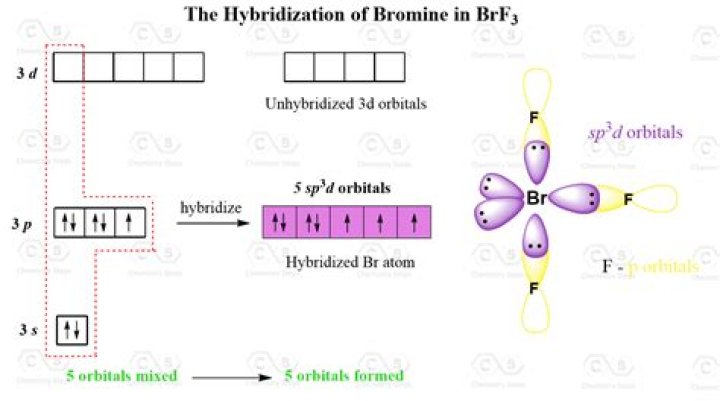
Name of the MoleculeBromine PentafluorideMolecular FormulaBrF5Hybridization Typesp3d2Bond Angle90oGeometrySquare Pyramidal
How many electron domains are on the central atom of BrCl3?
From the Lewis structure drawn above, we see that there are five electron domains. Two of them are the lone pairs on the Br atom and 3 of them are the single bonds between the Br and Cl atoms.
What is the correct Iupac name for BrCl3?
PubChem CID12597988Molecular FormulaBrCl3SynonymsBromine trichloride 12360-50-8 trichloro-lambda3-bromane EINECS 235-610-8 DTXSID60154053Molecular Weight186.26DatesModify 2021-12-11 Create 2007-02-08
What is the molecular geometry of xef4?
XeF4 Molecular Geometry And Bond Angles In order to achieve this, the lone pairs lie in a perpendicular plane in an octahedral arrangement opposite (180 degree) from each other. Therefore, XeF4 molecular geometry is square planar.Which has sp2 hybridization?
Sulphur in SO2, is sp2-hybridised.
What is the hybridization of BF4?
BF3 has sp2− hybridisation, whereas BF4− has sp3− hybridisation.
What is the hybridization of the central atom in pf6?
The hybridization of the P atom in PF−6 P F 6 − is sp3d2 s p 3 d 2 and the geometry is octahedral.
What is the hybridisation of BCl3?
Hybridization of BCl3 (Boron Trichloride) The type of hybridization that occurs in BCl3 is sp2 hybridization.What is the hybridization of the central atom in sof4?
In sulphur hexafluoride, the central sulphur atom is attached to six fluorine atoms through six sigma bonds and there is no lone electron pair on sulphur atom. So, the steric number of sulphur is 6. Hence, sulphur atom is sp3d2 hybridized.
What is the hybridization around the bromine in BrO3?The total valence electron available for drawing the Bromate ion (BrO3-) lewis structure is 26. The hybridization in BrO3- is Sp3.
Article first time published onWhat is the hybridization of C in HCN?
In HCN molecule, the C atom includes sp-hybridized orbital, since it will combine with only two other atoms to form HCN.
Why is XeF4 a square planar?
For XeF4 , there is a plane of symmetry so it is non-polar. Since there are 4 bonds to the central atom and 2 lone pairs, the geometry is square planar.
How do you find the hybridization of XeF2?
XeF2 has 2 sigma bonds and 3 lone pairs of electrons. So its hybridization turns out to be sp3d. Its molecular geometry is Trigonal bipyramidal and its shape is linear as the lone pairs occupy the equatorial positions. The bond angles will be 90° and 180° in the plane of the molecule.
What is the hybridization of XeF2?
The hybridization of XeF2 (Xenon Difluoride) is an sp3d type.
What is the hybridization of phosphorus in the following molecule?
The central atom phosphorus is involved in sp3 hybridisation with three bond pairs and one lone pair.
Is BrCl3 a Lewis structure?
In the BrCl3 Lewis structure Bromine (Br) is the least electronegative atom and goes in the center of the Lewis structure. For the BrCl3 Lewis structure, you’ll need to put more than eight valence electrons on the Bromine atom. In the Lewis structure for BrCl3 there are a total of 28 valence electrons.
How do you find the hybridization?
- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
What is sp2 and sp3 hybridization?
sp hybridization occurs due to the mixing of one s and one p atomic orbital, sp2 hybridization is the mixing of one s and two p atomic orbitals and sp3 hybridization is the mixing of one s and three p atomic orbitals.
What is the hybridization of Se in sebr6?
Answer and Explanation: In hybridization, the atomic orbital of proper energy and symmetry will combine o form molecular orbitals. Here the one s, three p and two d atomic orbitals of Se combine forming six sp3d2 s p 3 d 2 molecular orbitals and six bromine atoms make six bonds with Se forming the entire molecule.
What is the hybridization of Sulphur in SF6?
The hybridization of SF6 is sp3d2 type. Just to describe the compound in brief, Sulphur Hexafluoride is a type of greenhouse gas which is colourless, odourless, non-toxic and non-flammable.
Is BF4 a sp3?
In BF4- the hybridization of Boron is sp3. The geometry of the molecule is tetrahedral and thus the F-B-F bond angle is 109.5° .
What is the hybridization of so4 2?
A The S in the SO42− ion has four electron pairs and has four bonded atoms, so the structure is tetrahedral. The sulfur must be sp3 hybridized to generate four S–O bonds.
What is hybridization of B in BF3 and BF4?
sp2,sp2.
Why does BCl3 have sp2 hybridization?
Each chlorine atom uses its half filled p-orbital for the σ-bond formation. Thus the shape of BCl3 is trigonal planar with bond angles equal to 120°. Boron forms three σsp-p bonds with three chlorine atoms by using its half filled sp2 hybrid orbitals.
What is the hybridization of Br in bro4?
The hybridization of Br in BrO−4 B r O 4 − is sp2 s p 2 and it has tetrahedral structure.
Does BrO3 have a central atom with Sp3 hybridization?
In BrO3 – central metal atom is ‘Br’ and it has 3 -sigma bond and 1- lone pair present ,therefore it has sp3 hybridisation and trigonal shape and in ‘HOCl’ central metal atom is ‘O’ and it has 2- sigma bond and 2-lone pair therefore it has Sp3 hybridisation and angular shape.
What is the shape of HCN?
FormulaShape4.H3O+trigonal pyramidal5.HCNlinear6.CO2linear7.CCl4tetrahedral
How many atoms are in HCN?
HCN Lewis structure comprises three different atoms: Hydrogen, carbon, and nitrogen.
Which of the following molecule has sp3 hybridization?
MethaneThe methane molecule has four equal bonds. In hybridization, carbon’s 2s and three 2p orbitals combine into four identical orbitals, now called sp3 hybrids.