What is addition reaction example
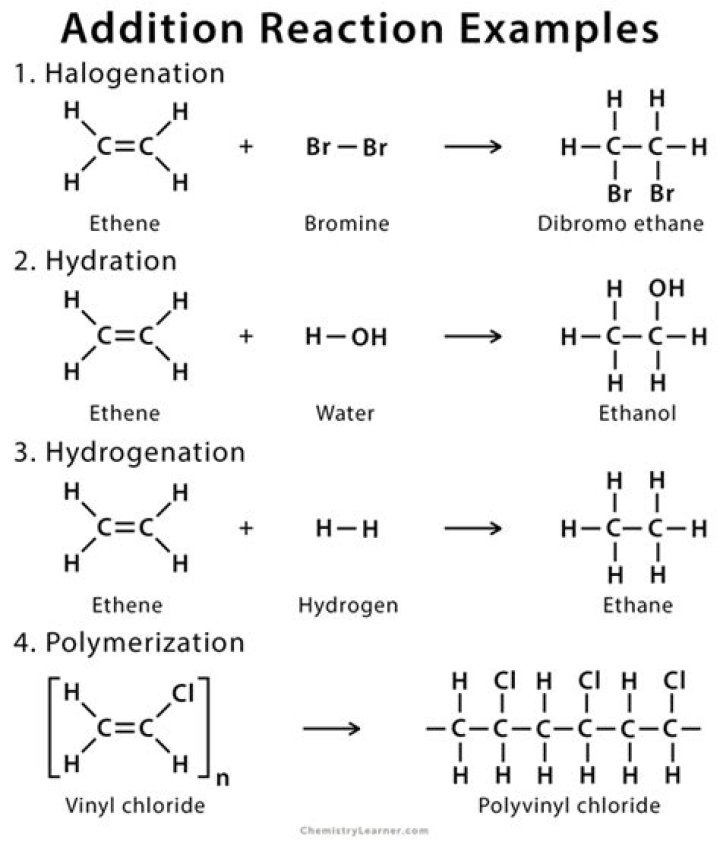
Examples of addition reactions that are common in alkene include halogenation, hydration, hydrohalogenation, hydrogenation, and polymerization. Alkyne can undergo addition reaction to give an alkene or its derivative.
What are the 4 types of addition reactions?
Addition Reaction – Electrophilic, Nucleophilic, Free-radical Addition Reaction with FAQs.
What is addition reaction Class 10?
An addition reaction occurs when two or more reactants combine to form a single product. Addition reactions occur with unsaturated compounds. The general equation for an addition reaction: A+BA+B →→ C.
What is the difference between additive and synergistic effect?
Additive effects are when the sum of the effect equals the two individual chemical effects combined. … Synergistic effects are when the sum of the effect is more than the two individual chemical effects combined.What is addition of alkene?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
What is E1 mechanism?
Unimolecular Elimination (E1) is a reaction in which the removal of an HX substituent results in the formation of a double bond. It is similar to a unimolecular nucleophilic substitution reaction (SN1) in various ways. One being the formation of a carbocation intermediate.
What is carbon addition?
Addition means the release of carbon into the atmosphere as carbon dioxide and methane by: respiration, excretion, decay. combustion eg the burning of fossil fuels. volcanic activity.
What is synergy and antagonism?
Thus, synergism is used to define a cumulative effect of multiple stressors that are greater than the additive sum of effects produced by the stressors acting in isolation; this contrasts with the term “antagonism,” used to define a cumulative effect that is less than additive (Hay et al.What is substitution chemistry?
A substitution reaction (also known as single displacement reaction or single substitution reaction) is a chemical reaction during which one functional group in a chemical compound is replaced by another functional group.
What is addition in pharmacology?The effect-addition model predicts that the combined action of two drugs is equal to the arithmetic sum of the individual effects. This is referred to as effect-additive and deviations from the predicted effects are described accordingly.
Article first time published onWhat is synergy in chemistry?
Synergy is commonly defined as the effect of two or more agents working in combination that is greater than the expected additive effect of said agents (Greco et al., 1996).
What is addition reaction for class 11th?
An Additional reaction is defined as the reaction where two compounds combine to form a new compound without losing any of its atoms. In this reaction weaker pi-bond is converted to form two new sigma bonds.
What are addition compounds?
An addition compound contains two or more simpler compounds that can be packed in a definite ratio into a crystal. The term covers donor-acceptor complexes (adducts) and a variety of lattice compounds.
What is substitution and addition reaction?
An addition reaction occurs when two or more reactants combine to form a single product. This product will contain all the atoms that were present in the reactants. … A substitution reaction occurs when an exchange of elements in the reactants takes place.
What is hydration of alkene?
Hydration of Alkenes The net addition of water to alkenes is known as hydration. The result involves breaking the pi bond in the alkene and an OH bond in water and the formation of a C-H bond and a C-OH bond.
What is a syn addition?
Syn addition: An addition reaction in which all new bonds are formed on the same face of the reactant molecule.
Why are alkenes reactive?
The number of hydrogen atoms in an alkene is double the number of carbon atoms, so they have the general formula. Alkenes are unsaturated, meaning they contain a double bond . This bond is why the alkenes are more reactive than the alkanes .
What is aldehyde and ketone?
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. … If at least one of these substituents is hydrogen, the compound is an aldehyde. If neither is hydrogen, the compound is a ketone.
Is hydration an addition reaction?
Alkenes undergo an addition reaction with water in the presence of a catalyst to form an alcohol. This type of addition reaction is called hydration. The water is added directly to the carbon – carbon double bond.
How do you write benzene?
The chemical formula for benzene is C6H6, i.e it has 6 hydrogen- H atoms and six-carbon atoms and has an average mass of about 78.112. The structure has a six-carbon ring which is represented by a hexagon and it includes 3-double bonds. The carbon atoms are represented by a corner that is bonded to other atoms.
What is ß elimination reaction?
β-Elimination (beta-elimination): A chemical reaction in which atoms or groups are lost from adjacent atoms, resulting in a new pi bond: A-B-C-D —> A + B=C + D. One of atoms lost is usually (but not always) a proton. … The reaction follows Zaitsev’s rule, giving the more highly substituted alkene as the major product.
What is chlorination of methane?
If a mixture of methane and chlorine is exposed to a flame, it explodes – producing carbon and hydrogen chloride. … The reaction we are going to explore is a more gentle one between methane and chlorine in the presence of ultraviolet light – typically sunlight.
Which is faster E1 or E2?
Mechanistically, E2 reactions are concerted (and occur faster), whereas E1 reactions are stepwise (and occur slower and at a higher energy cost, generally). Due to E1’s mechanistic behavior, carbocation rearrangements can occur in the intermediate, such that the positive charge is relocated on the most stable carbon.
What is halogenation in chemistry?
Halogenation is a chemical reaction that involves the reaction of a compound with a halogen and results in the halogen being added to the compound (Zhou et al., 2012c).
Is evaporation a chemical reaction?
Melting, evaporation and condensation are examples of physical change, or change of state, and are distinct from changes that cause new materials to form through a chemical reaction.
What is math substitution?
Substitution is the name given to the process of swapping an algebraic letter for its value. Consider the expression 8 + 4. This can take on a range of values depending on what number actually is. If we are told = 5, we can work out the value of the expression by swapping the for the number 5.
What is additive toxicity?
Abstract. Mixtures of chemicals or pesticides may produce unexpected effects; some are hazardous and some are beneficial. A method was divised in which individual toxic contributions of chemicals are summed, and the additive toxicity is defined by an index for two or more chemicals in combination.
What is synergy in pharmacology?
Synergy is commonly defined as the effect of two or more agents working in combination that is greater than the expected additive effect of said agents (Greco et al., 1996). … Synergy is most often defined in relation to the realms of pharmacology and medicine.
What is additive interaction?
Additive interaction is defined as a deviation from additivity of the absolute effects of two risk factors as originally described by Rothman [43,44].
What is triage in pharmacovigilance?
Triage refers to the process of placing a potential adverse event report into one of three categories: 1) non-serious case; 2) serious case; or 3) no case (minimum criteria for an AE case are not fulfilled).
What is drug concentration?
Drug concentration in the compartment is. defined as the amount of drug in a given volume, such. as mg/L: 1-1. Volume of distribution (V) is an important indicator of the extent of drug distribution into body fluids and tis- sues.