What is Atom in chemistry class 9
Atoms are defined as “the basic building blocks of matter”. It is the smallest constituent unit of matter that posses the properties of the chemical element. Atoms don’t exist independently, instead, they form ions and molecules which further combine in large numbers to form matter that we see, feel and touch.
What is atom Class 9 Ncert?
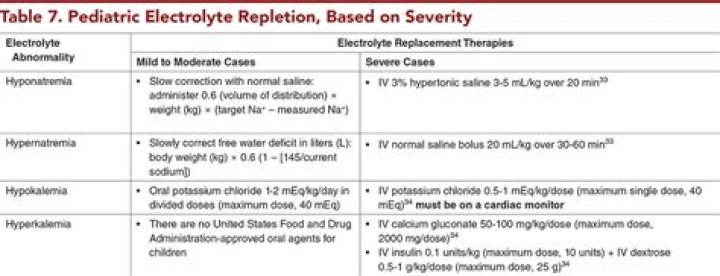
(i) All matter is made of very tiny particles called atoms. (ii) Atoms are indivisible particles, which cannot be created or destroyed in a chemical reaction. (iii) Atoms of a given element are identical in mass and chemical properties. (iv) Atoms of different elements have different masses and chemical properties.
What is atom Class 9 BYJU's?
Atoms are the building blocks of matter. … We know that Atoms are the smallest individual particle of a matter.
What is atom in chemistry short answer?
atom, smallest unit into which matter can be divided without the release of electrically charged particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element. As such, the atom is the basic building block of chemistry.What is atom in science class 8?
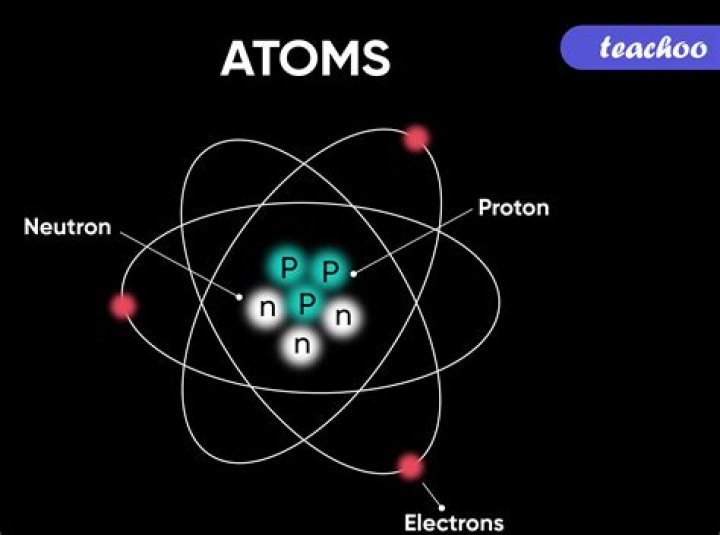
An atom is defined as the structural and functional unit of a matter. The term “atom” comes from the Greek word which means indivisible, because atoms are the smallest things in the universe and could not be divided. atoms are made up of three subatomic particles like protons, neutrons and electrons.
What is an atom in chemistry quizlet?
An atom is the smallest part of an element that can exist. Atoms are electrically neutral, with a positively charged, dense nucleus (filled with protons and neutrons) and surrounded by one or more electrons. Nucleus. the dense, positively charged mass located in the centre of an atom.
What is atom molecule and element?
An element is a substance that is made entirely of one type of atom. For example, the element sodium is made up of only sodium atoms. A molecule is a substance that contains two or more atoms chemically joined, such as H2,O2.
What are atom notes?
Atoms are made up of subatomic particles—protons, neutrons and electrons. o Protons and neutrons are in the nucleus of an atom. o Electrons occupy orbitals or electron clouds that surround the nucleus. • Protons are found in the nucleus of atoms and have an electric charge of +1.What is an atom in chemistry with examples?
Many atoms consist of a positively charged nucleus composed of protons and neutrons surrounded by a negatively charged cloud of electrons. At its most basic level, an atom is any particle of matter that contains at least one proton. … Here are some examples of atoms: Neon (Ne) Hydrogen (H)
What is an atom Class 8 answer?Complete answer: An atom consists of electrons, protons and neutrons. Electrons in an atom revolve around the nucleus in stationary orbits but protons and neutrons are present in the nucleus. Number of protons and number of electrons in a particular atom is the same and is known as its atomic number.
Article first time published onWhat is an atom in an element?
An atom consists of a tiny nucleus made up of protons and neutrons, on the order of 20,000 times smaller than the size of the atom. … A chemical element consists of those atoms with a specific number of protons in the nucleus; this number is called the atomic number.
Why is an atom an element?
An element is a substance that is made entirely from one type of atom. For example, the element hydrogen is made from atoms containing just one proton and one electron. … All isotopes of a particular element have the same number of protons, but can have different numbers of neutrons.
What are atoms made of?
Atoms are constructed of two types of elementary particles: electrons and quarks. Electrons occupy a space that surrounds an atom’s nucleus. Each electron has an electrical charge of -1. Quarks make up protons and neutrons, which, in turn, make up an atom’s nucleus.
What is an atom and its function?
An atom is the smallest unit of matter that retains all of the chemical properties of an element. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. For example, water is composed of hydrogen and oxygen atoms that have combined to form water molecules.
What is an atom in chemistry class 7?
Atom is the basic unit of matter. It constitutes the smallest particle of an element. In 1808, John Dalton proposed the Atomic theory which states that: Matter is made up of tiny particles called atoms. Atoms are indivisible and cannot be created or destroyed.
What is atom type?
Every atom is a chemical element, like hydrogen, iron or chlorine. Every element has cousins called isotopes. These have a different number of neutrons, but are otherwise the same. Having excess neutrons may make isotopes radioactive.
What is an atom answer in one sentence?
Atoms consist of a nucleus made of protons and neutrons orbited by electrons. Atoms are the basic units of matter and the defining structure of elements. The term “atom” comes from the Greek word for indivisible, because it was once thought that atoms were the smallest things in the universe and could not be divided.
What is an atom for kids?
The atom is the basic building block for all matter in the universe. Atoms are extremely small and are made up of a few even smaller particles. The basic particles that make up an atom are electrons, protons, and neutrons. Atoms fit together with other atoms to make up matter.
What is proton and neutron?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).
How are atoms formed?
Atoms were created after the Big Bang 13.7 billion years ago. As the hot, dense new universe cooled, conditions became suitable for quarks and electrons to form. Quarks came together to form protons and neutrons, and these particles combined into nuclei.
What is electrons made of?
Electrons are fundamental particles so they cannot be decomposed into constituents. They are therefore not made or composed. An electron acts as a point charge and a point mass.
Who discovered the atom?
The idea that everything is made of atoms was pioneered by John Dalton (1766-1844) in a book he published in 1808. He is sometimes called the “father” of atomic theory, but judging from this photo on the right “grandfather” might be a better term.
What is called molecule?
molecule, a group of two or more atoms that form the smallest identifiable unit into which a pure substance can be divided and still retain the composition and chemical properties of that substance.
What is the main idea of atoms?
The main points of Dalton’s atomic theory are: Everything is composed of atoms, which are the indivisible building blocks of matter and cannot be destroyed. All atoms of an element are identical. The atoms of different elements vary in size and mass.
What is an atom Class 11?
Atom is the smallest particle of matter that can take part in a chemical reaction. Atom is made of electron, proton and neutrons. Atom is not capable of independent existence. Two or more atoms combine together to form molecules.