What is bond order in NO3 ion
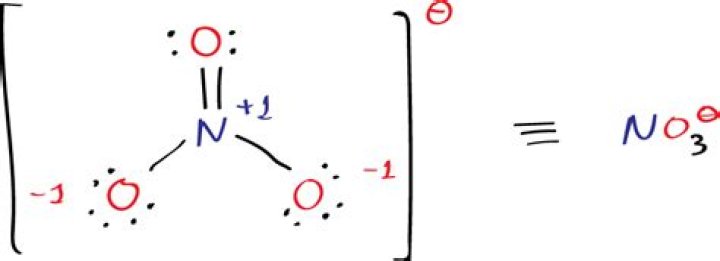
The Lewis structure of nitrate ion is unusual because it features 3 formal charges in that the nitrogen is quaternized and formally cationic, and 2 of the oxygen atoms have formal negative charges. Total number of bonds = 4. Number of bond groups between individual atoms= 3. ∴ Bond order = 4/3 = 1.33.
What is the average bond order for no?
Question: The average bond order is the number of bonds between two atoms taking into account resonance. Draw a Lewis structure for nitryl fluoride and answer the questions below. There are equivalent Lewis structures for NO_2F. The average N-O bond order is 1.33.
What is the bond order in the NO molecule?
So, the bond order of a nitric oxide molecule is 2.5.
What is the no bond order in NO2?
Each N-O bond order in NO2- is 1.5. Similarly in nitrate ion, NO3-, there are three equivalent resonance structures.How do you find bond order in no?
The Bond Order Formula can be defined as half of the difference between the number of electrons in bonding orbitals and antibonding orbitals. a = Number of electrons in bonding molecular orbitals. b = Number of electrons in antibonding molecular orbitals. Bond order of NO+ is 3.
What is the bond order of Cl2?
The bond order of Cl2 is 1.
Which has a greater bond length NO3 or NO2?
There are more such pairs in NO3− than in NO2−. These pairs tend to repel each other and the bonding pairs, and NO3− requires more room, thus longer bonds, to accommodate the greater number of closely packed nonbonding electron pairs.
What is the resonance structure of no3?
In lewis structure of NO3- ion, there are three lone pairs (in the last shell) in two oxygen atom and that oxygen atoms. Also, those two oxygen atoms has a -1 charge. There is another oxygen atom. That oxygen atom is connected to the nitrogen atom by a double bond has two lone pairs in its last shell.What is the average no bond order in NO2+?
Thus, NO2+ loses the 2b1 antibonding electron and the 3a1 bonding electron, and its bond order is around 2.5 .
What is bond order of b2?Answer: The bond order of B2 molecule is one.
Article first time published onWhat is bond order of NO molecule Mcq?
Bond order for both NO+ and NO− is 2.5.
Which of the following has 0 bond order?
Dihydrogen (H2) with an Electron in the Antibonding Orbital The electron configuration of dihelium: If the molecule He2 were to exist, the 4s electrons would have to fully occupy both the bonding and antibonding levels, giving a bond order of zero.
What is the bond order of li2 +?
The molecule Li2 is a stable molecule in the gas phase, with a bond order of one.
Why is the bond order of CO 3?
The bond order of CO is 3 as oxygen shares two of its electrons with carbon and makes a dative bond filling its a shell.
What is bond order in N2 CO and NO +?
(I) Bond order of CO and N2 are 3 and 3. Both CO and nitrogen are isoelectronic and contain 14 electrons each. Hence, they have same bond order.
Which has a shorter NO bond length NO2 or NO3?
Bond length increases with decreasing bond order. The nitrogen–oxygen bond order in NO3- is less than the nitrogen–oxygen bond order in NO2- (1.3 versus 1.5, respectively). Therefore, NO3- has longer nitrogen–oxygen bonds than does NO2-.
How should the NO bond lengths compare in the NO3 and NO2 ions?
bond lengths. Both N–O bonds in NO2 are identical (bond length 120 pm) and are intermediate between N-O and N =O. bond lengths. Both of the N-O bond distances in the nitrite ion, NO2, are 124 pm.
Which ion has the longest NO bond?
Out of NO+, NO2-, NO3- which has the longest NO bond and which has the shortest NO bond? The bond order for NO+ is 3 electrons/1 bonding group = 3. The bond order for NO−2 is 3 electrons/2 bonding groups = 3/2 = 1.5.
What is Cl2 bond length?
Intermolecular distances in weakly bound complexes have been usually interpreted in terms of van der Waals radii of atoms/molecules. … Chlorine bond radius for ClF and Cl2 are determined to be 1.28±0.11 Å and 1.55±0.07 Å, respectively.
What is electronic configuration of Cl2?
2.1 Chlorine (Cl2) The electronic configuration of chlorine is: 1s2 2s2 2p6 3s2 3p5. Alternatively this can be written [Ne] 3s2 3p5 in other words the core electrons are 1s2 2s2 2p6 corresponding to the electronic configuration of the noble gas neon.
What is the bond angle of NO3 minus?
Thus, NO3- molecular geometry is trigonal planar and is slightly bent. The bond angle is 120°.
Why are the no bonds in NO2 the same length?
The N−O bonds in the NO2− ion are equal in length, whereas they are unequal in HNO2. Explain. Since the nitrogen dioxide ion has resonance, the N−O bonds are equal as resonance is in reality a hybrid of all of the possible structures for a certain molecule.
Why is there a double bond in no3?
The nitrate ion is formed by the loss of the hydrogen ion, and so its structure is: Around the central nitrogen there are 4 pairs of shared electrons, and no remaining lone pair. The original lone pair has now become a bonding pair. Two of those pairs make up a double bond.
What is the charge of no3?
For example, NO−3 is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall 1− charge.
What is the total number of bonds in the Lewis structure of the nitrate ion?
The nitrate ion, according to its Lewis diagram, has two types of nitrogen-oxygen bonds, one double bond and two single bonds, suggesting that one nitrogen-oxygen bond in the nitrate ion is shorter and stronger than each of the other two.
Is Li2 paramagnetic or diamagnetic?
Li2 only has 2 electrons. If you draw the MO diagram, they should both be in the sigma 2s bonding orbital. They are both paired, so it is diamagnetic.
Which molecule does not exist due to zero bond order?
7. Which of the following molecule does not exist due to its zero bond order? Explanation: Molecular orbital electronic configuration of He2 molecule = (σ1s)2 (σ*1s)2. Bond order = 0, so He2 molecule does not exist.
Which of the following molecule is not Homonuclear?
Which of the following molecule is not homonuclear? Explanation: NO is Heteronuclear diatomic molecule in which atomic number of nitrogen is 7 and that of oxygen is 8 i.e., total number of electrons = 15.
Which of the following molecule does not exist?
Explanation: The electronic configuration of He2 is σ1s^2σ*1s^2. Here NA = NB (where NA as the number of anti-bonding molecular orbitals and NB as the number of bonding molecular orbitals). So the molecule He2 doesn’t exist.
How many bond does b2 have?
B2 possesses two different kinds of σ bonds: a strong, short bond and a longer, weak bond. In B2+, no π bond exists, only two σ bonds.
How many electrons does Li2 have?
That means there are 3 electrons in a lithium atom. Looking at the picture, you can see there are two electrons in shell one and only one in shell two.



