What is called sublimation answer
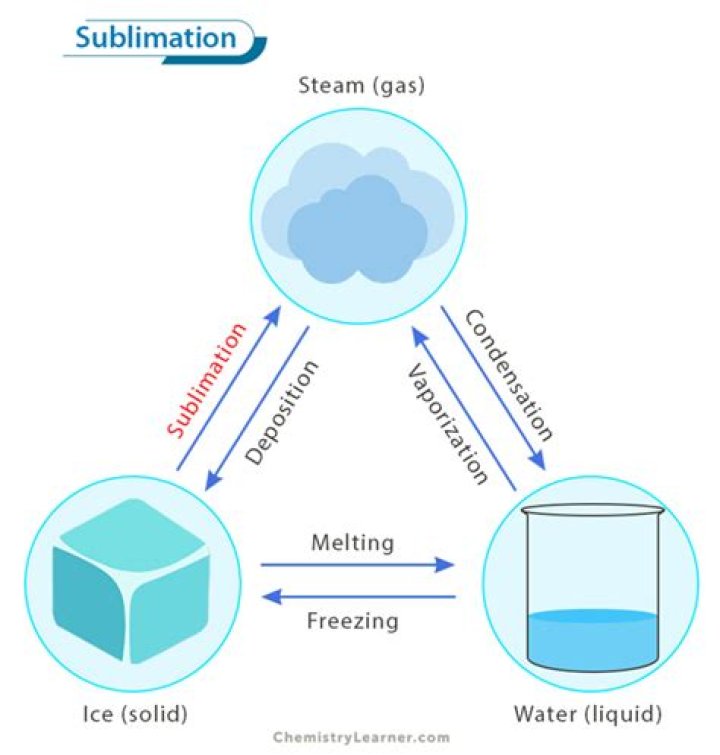
Sublimation is the transition of a substance directly from the solid to the gas state, without passing through the liquid state.
What is sublimation with example?
sublimation, in physics, conversion of a substance from the solid to the gaseous state without its becoming liquid. An example is the vaporization of frozen carbon dioxide (dry ice) at ordinary atmospheric pressure and temperature. The phenomenon is the result of vapour pressure and temperature relationships.
What is mean by sublimation in chemistry?
Sublimation is the transition of a substance directly from the solid phase to the gas phase without passing through the intermediate liquid phase (Table 4.8, Fig. 4.2). Sublimation is an endothermic phase transition that occurs at temperatures and pressures below the triple point of a chemical in the phase diagram.
What is sublimation class 9th?
sublimation. The Changing of a solid directly into vapours on heating,and of vapours into solid on cooling is called as sublimation. The solid substance which undergoes sublimation is called sublime.What is sublimation class 6th answer?
Sublimation means substance directly converted in gaseous form. Those substances convert in gaseous form this is called as sublimate. Sublimation is also one kind of separation technique which we can use to separate solid-solid homogeneous mixture which is difficult to separate from one another.
What is liquid to gas called?
vaporization, conversion of a substance from the liquid or solid phase into the gaseous (vapour) phase. If conditions allow the formation of vapour bubbles within a liquid, the vaporization process is called boiling.
What is sublimation 11th class?
Hint: Sublimation can be defined as the transition of a substance from solid to gaseous phase without forming the liquid phase.
What is sublimation Vedantu?
Sublimation is defined as the change or transition from the solid phase into the gas phase without entering the liquid phase. … The solid substance that undergoes the change to form gas is called Sublime. The solid obtained by cooling of the vapors is called as sublimate.What is sublimation class 4th?
The term sublimation is the passage or the transformation or conversion that substances undergo when passing from one state to another, for example from a solid substance to gas. We can define sublimation as the transition of a substance from the solid phase to the gaseous phase without changing into the liquid phase.
Which substances can sublimate?Familiar substances that sublime readily include iodine (shown below), dry ice (shown below), menthol, and camphor. Sublimation is occasionally used in the laboratory as a method for purification of solids, for example, with caffeine.
Article first time published onWhat is mixture Class 9?
A mixture is a substance which consist of two or more elements or compounds not chemically combined together. For Example Air is a mixture of gases like oxygen,nitrogen,argon,carbon dioxide etc. … The various substance present in a mixture are called as its constituents or components.
What is sublimation Ncert?
The phenomenon of change of solid directly to gas or conversion of gas directly to liquid without changing into liquid state is called sublimation.
What is sublimation 8th class?
Sublimation is the process in which a solid changes directly to a gas without going through the liquid state. Solid carbon dioxide is an example of a substance that undergoes sublimation.
How does ice sublimate?
When ice turns directly into water vapor without first transitioning into a liquid, it is referred to as “sublimation.” In fact, any solid that turns into a vapor without going through the liquid phase can be said to sublime or “sublimate.”
What is the 4th state of matter?
Plasma, the fourth state of matter (beyond the conventional solids, liquids and gases), is an ionized gas consisting of approximately equal numbers of positively and negatively charged particles.
What is liquid to solid called?
Freezing occurs when a liquid is cooled and turns to a solid. … This is called freezing and occurs at the same temperature as melting. Hence, the melting point and freezing point of a substance are the same temperature.
What causes vaporization?
Vaporization on Atomic Level Both types of vaporization occur when the temperature reaches a certain level, whether on the surface or throughout the liquid. … As these bonds are broken, the molecules and atoms separate and spread out, which causes them to vaporize, or turn into a gas.
What is sublimation BYJU's?
Sublimation is the transition of a substance directly from the solid to the gas phase, without passing through the intermediate liquid phase.
What is sublimation of camphor?
Camphor converts directly into its gaseous state without converting into liquid state. This property of camphor helps to separate it from NaCl. The process is called sublimation. … So when a mixture of sodium chloride and camphor is heated, camphor gets sublime and gets directly converted into vapor form.
What is the remainder product of sublimation called?
d) Sublimate. Explanation: The solid remaining is termed as sublimate. 7.
Which industry uses sublimation?
Apparel and garments industry is one of the largest end-use sector for sublimation printing. Frequently changing fashion trends are also expected to contribute significantly to the global sublimation printing equipment market.
Is dry ice sublimation?
Sublimation and the water cycle: “Dry ice” is actually solid, frozen carbon dioxide, which happens to sublimate, or turn to gas, at a chilly -78.5 °C (-109.3°F).
What is Fullform mixture?
In chemistry, a mixture is a material made up of two or more different chemical substance/substances which are not chemically combined. A mixture is the physical combination of two or more substances in which the identities are retained and are mixed in the form of solutions, suspensions and colloids.
What is solvent and solute?
The solute is the substance that is being dissolved, while the solvent is the dissolving medium. Solutions can be formed with many different types and forms of solutes and solvents.
What is solvent class9?
The liquid in which solute is dissolved is called SOLVENT. For Ex:Salt Solution(Salt and water) Salt is a solute and water is a solvent. Solute particles are lesser in amount and are called dispersed particles. Solvent particles are greater in amount and are called dispersed medium.
What is evaporation answer?
Evaporation is the process by which water changes from a liquid to a gas or vapor. Evaporation is the primary pathway that water moves from the liquid state back into the water cycle as atmospheric water vapor.