What is helium number of neutrons
Most Helium atoms have two neutrons in addition to the protons. In its neutral state, Helium has two electrons in orbit about the nucleus. Model of a helium atom’s nucleus with two protons and two neutrons.
How many protons and electrons are in helium?
A helium atom has 2 protons, 2 electrons and a mass which is about 4 times that of a hydrogen atom.
How do you find a proton number?
Finding the Number of Protons The atomic number is located above the element’s symbol. Since oxygen has an atomic number of eight, there must be eight protons total. Moreover, the number of protons never changes for an element.
What is the proton and neutron of helium?
A helium atom has 2 protons and 2 neutrons in the nucleus.How many protons are in a helium ion?
A helium atom has two electrons in an energy level outside the nucleus. The atom is neutral as it has two positive protons and two negative electrons. A helium atom that has lost or gained an electron is a helium ion.
How many protons does helium 6 have?
* Helium-6 is the simplest nucleus with a “halo” — two loosely bound neutrons in an orbit around a compact core formed by two protons and two neutrons, also known as an alpha particle.
How many protons does helium 5 have?
Helium-5 is an unstable isotope of Helium. It has three neutrons. Make a He-5 nucleus with three neutrons and two protons.
What is the proton electron and neutron of helium?
NameHeliumSymbolHeProtons2Neutrons2Electrons2What is proton and neutron?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).
What is the proton symbol?ParticleSymbolChargeelectrone–1protonp++1neutronno0
Article first time published onWhat is meant by proton number?
The proton number (Z) of an atom is the number of protons it contains. All the atoms of a particular element have the same proton number (number of protons). … The nucleon number (or mass number) of an atom is the total number of protons and neutrons it contains.
Where is the proton located?
Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus.
What is helium composed of?
Helium is composed of two electrons, two protons, and usually two neutrons. It is a colorless, odorless, inert gas. Helium is a byproduct of both fusion and fission.
What is helium-4 made up of?
Helium-4 is made of 2 protons (the part of an atomic nucleus with a positive charge), 2 neutrons (the part of an atomic nucleus with no charge) and 2 electrons (the part of an atom that goes around the nucleus with a negative charge). Many helium-4 on Earth is produced from the alpha decay of heavy elements.
Is helium a gas?
helium (He), chemical element, inert gas of Group 18 (noble gases) of the periodic table. The second lightest element (only hydrogen is lighter), helium is a colourless, odourless, and tasteless gas that becomes liquid at −268.9 °C (−452 °F).
How many protons does tritium have?
tritium, (T, or 3H), the isotope of hydrogen with atomic weight of approximately 3. Its nucleus, consisting of one proton and two neutrons, has triple the mass of the nucleus of ordinary hydrogen.
Why is hydrogen a proton?
A hydrogen ion is called a proton because hydrogen atoms which have only one electron lose that one electron to become an ion leaving only one…
Who discovered proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
Why is helium a positive ion?
Element with a nucleus that loses excess energy by spontaneously releasing an alpha particle, which is a positively charged helium ion.
Is helium ionic or covalent?
The extremely stable noble gasses, including helium, neon, argon, krypton, xenon and radon, are all also nonmetal covalent elements. These elements form bonds with one another by sharing electrons to form compounds.
How many neutrons are in CA?
Johnson Z. In calcium-40, there are 20 neutrons.
How many protons are there in helium?
The nucleus of every helium atom contains two protons, but, as is the case with all elements, isotopes of helium exist. The known isotopes of helium contain from one to six neutrons, so their mass numbers range from three to eight.
How many protons does boron have?
When scientists count four protons in an atom, they know it’s a beryllium atom. An atom with three protons is a lithium atom, an atom with five protons is a boron atom, an atom with six protons is a carbon atom… the list goes on.
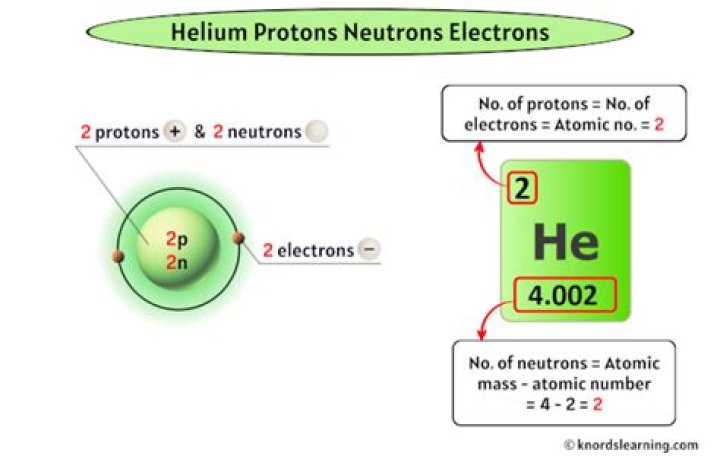
How many protons neutrons and electrons are in helium?
The nucleus of a helium atom contains two protons and two neutrons, and the helium atom has two electrons.
What element contains 7 protons?
If it has 7 protons, that’s nitrogen. But the logic of the periodic table also relies upon the number and organisation of electrons in an atom. Let’s take chlorine as an example. It is positioned in group 7 of the periodic table because it has seven electrons in the outermost shell of electrons.
How many neutrons does helium-3 have?
superfluid research … stable isotopes of helium are helium-3 (or 3He), with two protons and one neutron, and helium-4 (or 4He), with two protons and two neutrons.
How many protons and electrons does boron have?
Definition: Boron is the element with atomic number 5. Each boron atom has five protons in its nucleus (atomic core), and five electrons.
How many protons does each element have?
ElementLithiumSymbolLiNumber of Electrons in Each ShellFirst2Second1
What charge is a proton?
Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite. Neutrons have no charge.
What is proton neutron and electron?
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).
What is proton and electron?
Electrons are a type of subatomic particle with a negative charge. Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. … The positive charge on a proton is equal in magnitude to the negative charge on an electron.


