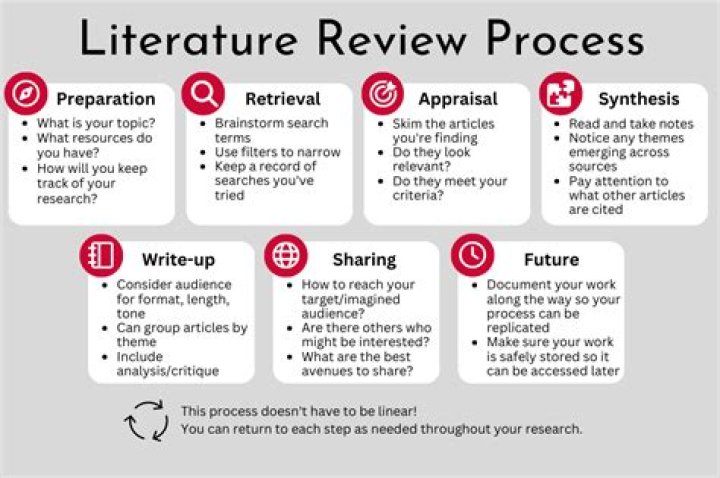
What is meant by genotoxicity

Genotoxicity is defined as a destructive effect affecting the integrity of the genetic material of the cells (DNA, RNA).
What causes genotoxicity?
Causes for this structure are mitotic loss of acentric chromosomal fragments (clastogenicity), mechanical problems from chromosomal breakage and exchange, mitotic loss of chromosomes (aneugenicity), and apoptosis.
How do you calculate genotoxicity?
The most commonly applied methods for detecting genotoxicity include the bacterial Ames test, DNA strand break measurements in cells (e.g. comet assay, alkaline unwinding and hydroxyapatite chromatography, alkaline elution), and cytogenetic assays (micronucleus and chromosomal aberration assays, including the use of …
What is a genotoxicity study?
Genotoxicity tests can be defined as in vitro and in vivo tests designed to detect compounds that induce genetic damage by various mechanisms. These tests enable hazard identification with respect to damage to DNA and its fixation.What is genotoxicity and mutagenicity?
The genetic change is referred to as a mutation and the agent causing the change as a mutagen. Genotoxicity is similar to mutagenicity except that genotoxic effects are not necessarily always associated with mutations. All mutagens are genotoxic, however, not all genotoxic substances are mutagenic.
What is genotoxicity Slideshare?
continued Genotoxicity describes the property of chemical agents that damages the genetic information within a cell causing mutations, which may lead to cancer The alteration can have direct or indirect effects on the DNA: the induction of mutations and direct DNA damage leading to mutations.
Why is genotoxicity important?
Genotoxicity testing is an important part of preclinical safety assessment of new drugs and is required prior to Phase I/II clinical trials. It is designed to detect genetic damage such as gene mutations and chromosomal aberration, which may be reflected in tumorigenic or heritable mutation potential of the drug.
Is chemotherapy genotoxic?
Genotoxic drugs are chemotherapy agents that affect nucleic acids and alter their function. These drugs may directly bind to DNA or they may indirectly lead to DNA damage by affecting enzymes involved in DNA replication.What are genotoxic carcinogens?
Genotoxic carcinogens are chemicals that exert carcinogenicity via the induction of mutations. Owing to their DNA interaction properties, there is thought to be no safe exposure threshold or dose. Genotoxic carcinogens are regulated under the assumption that they pose a cancer risk for humans, even at very low doses.
How do you test for Carcinogenity?Mutagenicity/genotoxicity assays are the most commonly used in vitro test systems to predict carcinogenicity. Mutagenicity refers to the induction of transmissible changes in the structure of the genetic material of cells or organisms (Maurici, et al., 2005b). Mutations may involve a single gene or a group of genes.
Article first time published onAre vaccines genotoxic?
Data on the effects of immunobiological preparations, including vaccines, employed for preventive mass immunization on certain hereditary structures are reviewed. Many of these preparations are genotoxic. Prospects for diminishing the genetic risks of vaccinations are discussed.
What are genotoxic agents?
A genotoxic agent is a chemical or another agent that damages cellular DNA, resulting in mutations or cancer. Toxic to the genome! Genotoxic substances are known to be potentially mutagenic or carcinogenic when inhaled, ingested or penetrate the skin.
What is fate and purge study?
Fate and purge studies challenge the process by deliberately spiking known concentrations of impurities to determine how well they are purged from the process.
What is TTC value?
TTC values are numbers that describe generic human chronic exposure thresholds that have been established by grouping experimental toxicity data from animal bioassays.
Which of the following is a test for mutagenicity?
The Ames test, an in vitro gene mutation study in bacteria, is the basic test for mutagenicity. Positive result within in vitro tests, is followed by in vivo mutagenicity studies for further confirmation.
What is Clastogenic agent?
A clastogen is a mutagenic agent that disturbs normal DNA related processes or directly causes DNA strand breakages, thus causing the deletion, insertion, or rearrangement of entire chromosome sections. These processes are a form of mutagenesis which if left unrepaired, or improperly repaired, can lead to cancer.
What are two examples of carcinogens?
Carcinogens are substances or exposures that can cause cancer. Examples include home and workplace chemicals, environmental or medical radiation, smoke, and even some viruses and medications.
Which are genotoxic impurities?
Regulatory landscape Per the International Council for Harmonization (ICH) S2 (R1) Guideline, genotoxic impurities can be broadly defined as impurities that have been demonstrated to cause deleterious changes in the genetic material regardless of the mechanism.
What is Toxicology Slideshare?
Toxicology • The study of poisons • Poisons are chemical/physical agents that produce adverse responses in biological organisms Any substance can be toxic if introduced in a dose capable of disturbing the normal physiological homeostasis of the exposed body.
How do you do a comet assay?
Comet assay utilizes single cells to measure DNA damage. First, cells are embedded into agarose and then placed onto a slide. The slide is then immersed into lysis solution to break open the cell membrane. After the cells are lysed, DNA is denatured under neutral or alkaline conditions and run through electrophoresis.
How do genotoxic carcinogens work?
The term “genotoxic carcinogen” indicates a chemical capable of producing cancer by directly altering the genetic material of target cells, while “non-genotoxic carcinogen” represents a chemical capable of producing cancer by some secondary mechanism not related to direct gene damage.
What are 3 types of mutagens?
Mutagens can be physical mutagens, chemical mutagens, or biological mutagens. The ability of a substance to induce the alterations in the base pairs of DNA or mutation is known as mutagenicity.
What are the 6 classes of chemotherapy drugs?
- Normal cell cycle. …
- Alkylating agents. …
- Antimetabolites. …
- Anthracyclines. …
- Topoisomerase inhibitors. …
- Plant alkaloids. …
- Corticosteroids.
What are known carcinogens?
- Acetaldehyde (from consuming alcoholic beverages)
- Acheson process, occupational exposure associated with.
- Acid mists, strong inorganic.
- Aflatoxins.
- Alcoholic beverages.
- Aluminum production.
- 4-Aminobiphenyl.
- Areca nut.
What makes a carcinogen?
A carcinogen is an agent with the capacity to cause cancer in humans. Carcinogens may be natural, such as aflatoxin, which is produced by a fungus and sometimes found on stored grains, or manmade, such as asbestos or tobacco smoke. Carcinogens work by interacting with a cell’s DNA and inducing genetic mutations.
What are the levels of carcinogens?
- Group 1: Carcinogenic to humans.
- Group 2A: Probably carcinogenic to humans.
- Group 2B: Possibly carcinogenic to humans.
- Group 3: Not classifiable as to its carcinogenicity in humans.
How do you manufacture mRNA vaccines?
Large scale production of mRNA vaccines consists in a 1 or 2-step in vitro reaction followed by a purification platform with multiple steps that can include Dnase digestion, precipitation, chromatography or tangential flow filtration.
What does the ICH do?
The ICH facilitates international electronic communication through the provision of Electronic Standards for the Transfer of Regulatory Information (ESTRI), giving the Electronic Common Technical Document (eCTD), allowing for the electronic submission of the Common Technical Document from applicant to regulator.
What are the four main components of pharmacovigilance?
Core Capabilities: Pharmacovigilance delivers four primary capabilities to pharmaceutical companies: • Adverse Event Case Management including expedited reporting; • Aggregate Reporting; • Signal Intelligence; and • Risk Management.
What is key starting material?
Notes: Key Starting Material (KSM) term is used for intermediates in pharmaceutical industry. These are building blocks of drug industry. India imports KSM for many medicines from China and due to Coronavirus fears, the drug industry of the country is facing recession concerns.