What is operational qualification
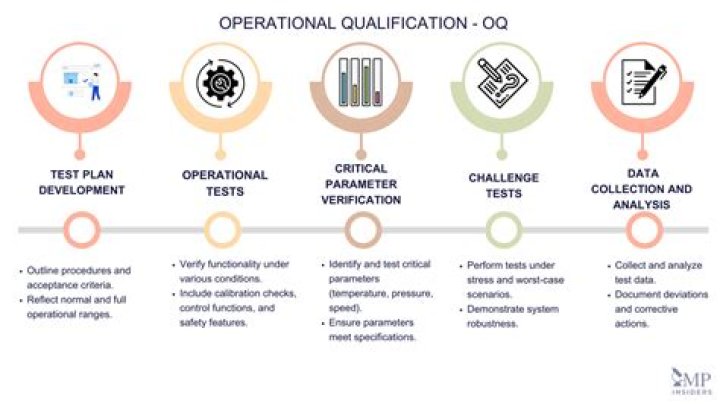
Operational Qualification, or OQ, is an essential process during the development of equipment often used by pharmaceutical companies. OQ can simply be defined as a series of tests which ensure that equipment and its sub-systems will operate within their specified limits consistently and dependably.
What is the different between OQ and PQ?
IQ, OQ, and PQ are sequential activities that manufacturers carry out to validate their manufacturing processes. IQ stands for Installation Qualification, OQ for Operational Qualification, and PQ for Performance Qualification.
What comes first OQ or PQ?
The Completion of OQ Leads to Performance Qualification (PQ) During this phase you will generate evidence that your process will consistently produce an acceptable product under normal operating conditions over the long-term.
What is a performance qualification?
Performance Qualifications are a collection of test cases used to verify that a system performs as expected under simulated real-world conditions. The performance qualification tests requirements defined in the User Requirements Specification (or possibly the Functional Requirements Specification).What is machine Qualification?
Machinery validation or Qualification is a mandatory requirement when ever a machine is used in a way that can affect the quality, safety, efficacy of a controlled product or jeopardize the integrity of any predicated data, relating to that product.
What is the purpose of CGMPs?
CGMPs provide for systems that assure proper design, monitoring, and control of manufacturing processes and facilities. Adherence to the CGMP regulations assures the identity, strength, quality, and purity of drug products by requiring that manufacturers of medications adequately control manufacturing operations.
What is operational Qualification in analytical chemistry?
Operational qualification (OQ) is the process of testing to ensure that the individual and combined systems function to meet agreed performance criteria and to check how the result of testing is recorded. The purpose is to ensure that all the dynamic attributes comply with the original design.
What is the difference between prospective and concurrent process validation?
The Difference Between Prospective, Concurrent and Retrospective Validation. … Prospective validation occurs before the system is used in production, concurrent validation occurs simultaneously with production, and retrospective validation occurs after production use has occurred.What does installation qualification mean?
Installation Qualification is a documented process that verifies that critical pieces of equipment, piping, software or instruments that directly impact product quality have been; Properly delivered. Correctly installed. Configured according to standards set by the manufacturer or by an approved installation checklist.
How do you write a performance qualification?Prepare a Performance Qualification Report: This should include: date study initiated; date completed; observations made; problems encountered; completeness of information collected; summary of deviation report; results of any tests; do results meet acceptance criteria; location of original data; other information …
Article first time published onWhat is IQ in validation?
IQ stands for Installation Qualification. This is the first step in the validation process to ensure that the manufacturing process will live up to its expectations. In this phase you verify the equipment’s design features and make sure it was installed correctly.
What is the difference between calibration and validation?
Calibration ensures that instrument or measuring devices producing accurate results. Validation provides documented evidence that a process, equipment, method or system produces consistent results (in other words, it ensures that uniforms batches are produced).
What does OQ mean in English?
AcronymDefinitionOQOperational QualificationOQOperator Qualification (personnel qualification in the natural gas and liquid pipeline business)OQOutcome Questionnaire (psychiatry)OQOverall Quality (Star Wars Galaxies game)
What is GxP in pharmaceutical industry?
GxP is a collection of quality guidelines and regulations created to ensure that bio/pharmaceutical products are safe, meet their intended use, and adhere to quality processes during manufacturing, control, storage and distribution.
What is IOQ in manufacturing?
The meaning of IQ, OQ and PQ are installation qualification, operational qualification and performance qualification, respectively. They represent quality assurance protocols for each phase of the manufacturing process for pharmaceutical equipment.
How many stages are there in qualification?
The four qualification stages help determine if the process being tested is capable of operating in its specific environment. They include: Qualification (DQ) Installation Qualification (IQ)
What is qualification in manufacturing?
Qualification is the process of demonstrating that an entity or process is capable of meeting or exceeding the specified requirements [1]. Qualification includes activities which ensure that the nominal design and manufacturing process will meet or exceed the specified targets.
What is DQ and IQ?
DQ Design Qualification means showing that a piece of technology – a device, apparatus, machine or system – has a GMP-compliant design. IQ Installation Qualification means showing it is set up, connected and installed as planned. OQ Operational Qualification means showing it works as intended in all respects.
What is DQ in pharmacy?
Design qualification is defined as a verification process on the design to meet particular requirements relating to the quality of pharmaceuticals and manufacturing practices. …
What is IQ in analytical chemistry?
Installation Qualification (IQ) is the first step in the qualification of new equipment. It is a documented process which verifies that all parts and pieces have been received and that an instrument has been installed and configured according to the manufacturer’s specifications or installation checklist.
What are the 3 importance of GMP?
GMP ensures that companies execute consistent procedures within safe environments. Hence, it prevents contamination, recalls, and loss of profit. GMP comes with strict protocols that lessen the risk of manufacturing errors. In the same way, companies can sustain efficient systems and processes to produce safe goods.
How do I become GMP certified?
- Application. This is the initial step to get the GMP certification; the application covers some essential data of the organization. …
- Review of Application. …
- Analysis & Agreement. …
- Documentation Review. …
- Granting Of Certification. …
- Surveillance Audit.
What is difference between GMP and cGMP?
cGMP is the advance version of the GMP because cGMP states for current GMP guidelines those are updated periodically by the pharmaceutical regulatory agencies for GMP inspection. … GMP ensures that the product is exactly what the product claims to be and that it was created by the standard guideline set known as GMP.
Why installation qualification is important?
The Installation Qualification Protocol verifies the proper installation and configuration of a System. This can include ensuring that necessary files have been loaded, equipment has been installed, the necessary procedures have been approved, or the appropriate personnel have been trained.
What is the objective of installation qualification?
The purpose of the installation qualification is to demonstrate that all the critical components of process equipment and support installations have been installed appropriately and are installed to the respective manufacturer’s or supplier’s requirements.
What should be included in an installation qualification?
- Location of install and necessary floor space.
- Documentation any and all computer-controlled instrumentation.
- Gathering all manuals and certifications.
- Properly unpacking and cross-checking instruments.
- Examining instruments and components for damage.
Why do you need 3 batches for validation?
Consideration of validation batches fewer than three will require more statistical and scientific data to prove the consistency of process to meet quality standards. … Therefore, minimum three consecutive batches are evaluated for validation of manufacturing process and cleaning procedures.
Why are there 3 batches for process validation?
As we know currently there are no guidelines available for selecting the minimum or maximum number of batches so we can also take 4 or 5 batches. As the number of batches is increased the cost of the process and time required is increased so all pharmaceutical companies usually select 3 batches for validation.
What is retrospective qualification?
A Retrospective Qualification is qualification of a system already in use based upon accumulated historical data’s conformance to predetermined acceptance criteria.
What is DQ IQ OQ in validation?
DQ Design Qualification means showing that a piece of technology – a device, apparatus, machine or system – has a GMP-compliant design. IQ Installation Qualification means showing it is set up, connected and installed as planned. OQ Operational Qualification means showing it works as intended in all respects.
What can be considered the worst case operational conditions?
A “worst case” condition or set of conditions are generally those parameters encompassing the upper and lower processing limits for a process and circumstances, within SOPs or batch records, which pose the greatest chance of product or process failure when compared to nominal conditions.



