What is syn and anti in chemistry
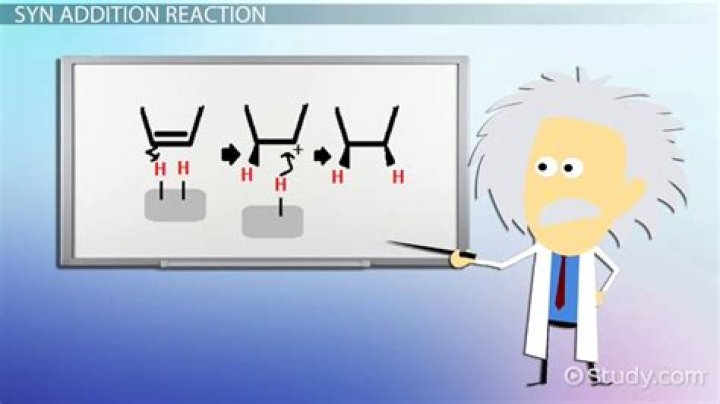
When two atoms are added to the same side, it’s a syn relationship; when two atoms are added to opposite sides it’s an anti relationship.
What is anti and syn?
The main difference between syn and anti addition is that syn addition involves the addition of substitutes to the same side of the double bond or triple bond whereas anti addition involves the addition of substitutes to the opposite sides of the double bond or triple bond.
Does syn mean CIS?
The difference is subtle: “cis” and “trans” implies that the orientation of the two groups is locked; you can’t convert cis to trans through rotation. The terms “syn” and “anti” imply similar relationships to “cis” and “trans”, but can also be applied to systems which are not locked.
How do you know if a reaction is syn or anti?
When both atoms/groups add to the same face it’s considered syn addition. When they add to opposite faces it’s considered anti addition. The difference between the two is determined by which atom the group added to (Mark vs Anti-Mark) or to which SIDE it adds (syn vs anti).What is syn addition in chemistry?
Syn addition is the addition of two substituents to the same side (or face) of a double bond or triple bond, resulting in a decrease in bond order but an increase in number of substituents. Generally the substrate will be an alkene or alkyne. … The classical example of this is bromination (any halogenation) of alkenes.
What does syn stand for?
AcronymDefinitionSYNSynchronizationSYNSynchronousSYNSynchronizeSYNSyndicate
How do you identify a syn addition?
An addition reaction of an alkene or an alkyne in which the net reaction is addition of two ligands to the multiple-bonded carbon atoms from the same face of the multiple bond is called a syn addition; one in which the net reaction is addition of the two ligands from the opposite faces of the multiple bond is called an …
What is Syn anti isomerism?
Definition of syn-anti isomerism : cis-trans isomerism in compounds (such as oximes, diazoates, and azo compounds) containing one or more carbon-to-nitrogen or nitrogen-to-nitrogen double bonds — compare benzaldoxime.What is Syn elimination?
In syn elimination, the base attacks the β-hydrogen on the same side as the leaving group. In anti elimination, the base attacks the β-hydrogen on the opposite side of the leaving group. It has been experimentally determined that E2 elimination occurs through an anti mechanism.
Is hydrogenation syn addition?Alkene hydrogenation is the syn-addition of hydrogen to an alkene, saturating the bond. The alkene reacts with hydrogen gas in the presence of a metal catalyst which allows the reaction to occur quickly.
Article first time published onWhich of these gives syn addition with alkenes?
Potassium permanganateKMnO4: diol addition in syn manner. , hydroboration followed by oxidation BH3−THF/H2O all these gives syn addition.
What is syn and anti geometrical isomers?
Hint: Syn means same side and anti means opposite side. Explanation: Geometrical isomerism is due to restricted rotation about the double bond. The prefixes syn and anti are used to denote geometrical isomerism. Syn: it is used to indicate that both H and OH are present on the same side of the double bond.
Is syn addition meso?
Organic Chemistry Many addition reactions to alkenes are stereoselective since one (set) of stereoisomers is formed from a starting material that is not stereoisomeric. … Therefore, it is a stereoselective reaction. A meso compound is formed when the cyclohexene undergoes a syn addition.
What is syn and anti hydroxylation?
Alkenes can be dihydroxylated by two different stereochemical pathways: anti-dihydroxylation or syn-dihydroxylation. The opening of epoxides follows the anti-dihydroxylation mechanism, while potassium permanganate or osmium tetroxide produce the syn-dihydroxylated products.
What is SYN Stereoselectivity?
Stereoselectivity. When an alkene undergoes addition, two new s bonds are formed. If we think of an alkene as having two faces, then the two new s bonds can either both form on the same face, which we call syn addition, or they can be formed on different faces which we call anti addition.
Is markovnikov syn or anti?
In the 3 membered ring pathway, the regiochemistry is also “Markovnikov”, the stereochemistry is trans (anti), and the reaction proceeds through a 3 membered ring intermediate.
What alkene reactions are syn?
A representative example of a syn addition to alkenes would be the hydroboration-oxidation reaction where the H and OH groups are adding to the same side of the double bond.
What does SYN mean in science?
a prefix occurring in loanwords from Greek, having the same function as co- (synthesis; synoptic); used, with the meaning “with,” “together,” in the formation of compound words (synsepalous) or “synthetic” in such compounds (syngas).
What does SYN mean in photosynthesis?
syn-, prefix. syn- comes from Greek, where it has the meaning “with; together. ” This meaning is found in such words as: idiosyncrasy, photosynthesis, synagogue, synchronize, synchronous, synonym, synthesis.
What does SYN mean in medical terms?
Syn-, Sym- = Together, Joined. Syn- is used to mean fixed or joined except when it is followed by the sound of “b”, “m”, “f”, “ph” or “p”. Then Sym- is used.
What is Syn elimination reaction?
The Ei mechanism (Elimination Internal/Intramolecular), also known as a thermal syn elimination or a pericyclic syn elimination, in organic chemistry is a special type of elimination reaction in which two vicinal substituents on an alkane framework leave simultaneously via a cyclic transition state to form an alkene in …
What does more substituted mean in organic chemistry?
More substituted carbon is one which forms greater no. of bonds with other carbon atoms. A less substituted carbon has less carbon atoms forming bonds to it. For example, a three degree carbon atom is more substituted carbon atom than a two degree one.
What is pyrolytic syn elimination?
Pyrolytic elimination or syn eliminations The term pyrolytic elimination literally means an elimination reaction occurring in the organic substrate due to the application of heat (Greek word pyr meaning fire).
Is hydrogenation a reduction?
Introduction. Addition of hydrogen to a carbon-carbon double bond to form an alkane is a reduction reaction that is also called catalytic hydrogenation. Hydrogenation of a double bond is a thermodynamically favorable reaction because it forms a more stable (lower energy) product.
Is hydration syn or anti?
The hydroboration mechanism has the elements of both hydrogenation and electrophilic addition and it is a stereospecific (syn addition), meaning that the hydroboration takes place on the same face of the double bond, this leads cis stereochemistry.
Is hydrogenation reaction syn or anti?
The greater the heat of hydrogenation, the less stable the alkene. In the addition both groups can add to the same face of the alkene (syn addition) or they can add to opposite faces (anti addition). Hydrogenation of alkenes is a syn addition.
Is bromination anti or syn?
Bromination of Alkenes Gives anti Products Although we saw that several key reactions of alkenes were consistent with this mechanism, it isn’t the case for all.
What is nomenclature system of geometrical isomerism?
E/Z System of Nomenclature of Geometrical Isomers. Cahn – Ingold – Prelog priority sequence can be usefully applied in giving names to such a structure. The group of highest priority, on both the double bonded carbon atom is first chosen. If the two group of highest priority are on the opposite side of the double bond.
Are diastereomers Nonsuperimposable?
Diastereomers are the stereomer compounds with molecules that are not mirrored images of one another and that are not superimposable. They are non-superimposable mirror images of each other.
What is the relationship between hexane and 3 Methylpentane?
Isomers are species of the same chemical formula, but with different connectivity. Both hexane and 3-methylpentane have formula of C6H14 , but the carbon atoms have different connectivity, and so are rightly classified as different chemical compounds even though they are isomeric.
Is Oxymercuration syn or anti?
Stereochemically, oxymercuration is an anti addition. As illustrated by the second step, the nucleophile cannot attack the carbon from the same face as the mercury ion because of steric hindrance.