What is temperature according to kinetic molecular theory
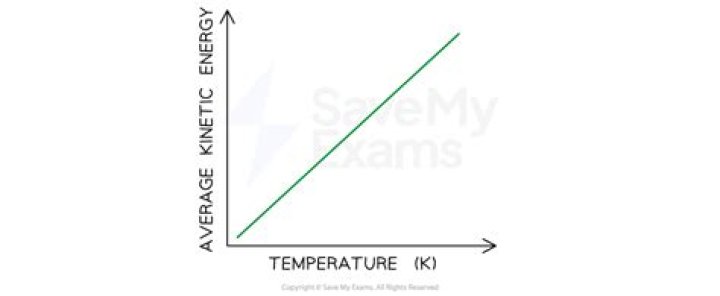
According to Kinetic Molecular Theory, an increase in temperature will increase the average kinetic energy of the molecules. … At a given temperature, the pressure of a container is determined by the number of times gas molecules strike the container walls.
What is temperature according to kinetic theory of gases?
The temperature of a gas is a measure of the mean kinetic energy of the gas. The molecules are in constant random motion, and there is an energy (mass x square of the velocity) associated with that motion. The higher the temperature, the greater the motion.
What is the relationship between temperature and kinetic energy in kinetic theory?
Temperature. Temperature is directly proportional to the average translational kinetic energy of molecules in an ideal gas.
What is the theory of temperature?
Temperature is a measure of the internal energy of a system, while heat is a measure of how energy is transferred from one system (or body) to another, or, how temperatures in one system are raised or lowered by interaction with another. This is roughly described by the kinetic theory, at least for gases and fluids.What is the relationship between temperature and molecular motion?
what is the relationship between molecular motion and temperature? The relationship between the two is that the higher the molecular motion is, the higher the temperature is.
What is temperature in terms of kinetic energy?
Temperature is a measure of the average kinetic energy of all the molecules in a gas. As the temperature and, therefore, kinetic energy, of a gas changes, the RMS speed of the gas molecules also changes.
What do you mean by mean kinetic temperature?
Mean kinetic temperature (MKT) is a simplified way of expressing the overall effect of temperature fluctuations during storage or transit of perishable goods. The MKT is widely used in the pharmaceutical industry.
Does kinetic energy depend on temperature?
As such, it can be concluded that the average kinetic energy of the molecules in a thermalized sample of gas depends only on the temperature. However, the average speed depends on the molecular mass. So, for a given temperature, light molecules will travel faster on average than heavier molecules.What is the relationship between Ke and temperature?
The Kelvin temperature of a substance is directly proportional to the average kinetic energy of the particles of the substance. For example, the particles in a sample of hydrogen gas at 200 K have twice the average kinetic energy as the particles in a hydrogen sample at 100 K.
What is kinetic interpretation of temperature derive kinetic energy in terms of temperature?The kinetic theory states that the average kinetic energy of gas molecules of an ideal gas is directly proportional to the absolute temperature of the molecules. … Such a kind of interpretation of temperature is called the kinetic interpretation of temperature.
Article first time published onWhat is temperature at molecular level?
At the molecular level, temperature is related to the random motions of the particles (atoms and molecules) in matter. Because there are different types of motion, the particles’ kinetic energy (energy of motion) can take different forms, and each form contributes to the total kinetic energy of the particles.
At which temperature does all molecular motion steps?
All molecular motion stops at 0 degrees Kelvin or -273.15 degrees Celsius.
What is the temperature at which all molecular motion stop?
The Kelvin Scale It is based on molecular motion, with the temperature of 0K, also known as absolute zero, being the point where all molecular motion ceases. The freezing point of water on the Kelvin scale is 273.15K, while the boiling point is 373.15K.
What is mean kinetic temperature in stability study?
As per ICH guidelines Mean Kinetic Temperature can be defined as a single calculated temperature which degrades the same amount of the drug as degraded by the different temperatures during the particular time period. …
How do you calculate temperature MKT?
- Convert °C to Kelvin by adding 273.15 to each reading.
- Calculate Delta H/(Gas Constant x Temperature Reading). …
- Calculate the sum of all results of Step 2.
- Divide the result of Step 3 by the number of readings.
How is temperature defined in terms of molecular action?
The greater a material’s temperature, the greater the thermal agitation of its constituent molecules (manifested both in linear motion and vibrational modes). It is natural for regions containing greater molecular kinetic energy to pass this energy to regions with less kinetic energy.
What is temperature called?
Degrees are used to indicate temperature. There are three types of temperature scales commonly used today: Celsius, Fahrenheit, and Kelvin. We are used to expressing temperature with degrees Fahrenheit (F). Scientists often use degrees Celsius (C), but the Kelvin (K) is the SI unit for temperature.
How do you find kinetic energy from temperature?
- Formula. K = (3/2) * (R / N) * T.
- Temperature (Kelvin)
- Gas Constant.
- Avogadro’s Number.
Why does temperature increase kinetic energy?
The kinetic energy increases as the particles move faster. The potential energy increases as the particles move farther apart. How are thermal energy and temperature related? When the temperature of an object increases, the average kinetic energy of its particles increases.
How is kinetic thermal temperature and heat related?
Heat is the sum of all the kinetic energies of all the molecules of an object, while temperature is the average kinetic energy of the molecules of an object.
How does temperature relate to kinetic energy quizlet?
temperature is the average kinetic energy of the particles of matter. the hotter something is the more kinetic energy it has. … temperature heats up the particles they have more kinetic energy and move and bounce into each other and spread out.
What is the difference between temperature and kinetic energy?
The key difference between kinetic energy and temperature is that kinetic energy refers to the property of a moving object and is the work needed to accelerate a body from its resting state, whereas temperature is the thermal energy present in all matter.
What is kinetic interpretation of temperature and hence define absolute zero of temperature?
The temperature of a body is the measure of the average kinetic energy of a body. As absolute zero is equal to 0 in the Kelvin scale, it is also known as the absolute scale.
What is the kinetic interpretation of absolute zero of temperature?
Absolute zero is the temperature at which all molecular motion ceases.
What is root mean square RMS speed and interpretation of temperature in kinetic theory?
(i) rms speed is directly proportional to square root of the temperature and inversely proportional to square root of mass of the molecule. … At a given temperature the molecules of lighter mass move faster on an average than the molecules with heavier masses.
What is the role of temperature in molecular movements?
In the kinetic theory of gasses, increasing the temperature of a gas increases in average kinetic energy of the molecules, causing increased motion. In the kinetic theory of gasses, increasing the temperature of a gas increases in average kinetic energy of the molecules, causing increased motion. …
How is temperature defined in chemistry?
Temperature is the property of matter which reflects the quantity of energy of motion of the component particles. It is a comparative measure of how hot or cold a material is. … It is the temperature where the thermal motion of particles is at its minimum (not the same as motionless).
What is temperature and heat?
Heat describes the transfer of thermal energy between molecules within a system and is measured in Joules. … Temperature describes the average kinetic energy of molecules within a material or system and is measured in Celsius (°C), Kelvin(K), Fahrenheit (°F), or Rankine (R).
Why does temperature remain constant during a phase transition?
During a change of the state of matter, the supplied energy is not used to increase the kinetic energy of the molecules, but to change the binding energies. Therefore, the temperature remains constant.
What do you mean by bracketing and Matrixing in stability?
Bracketing design in which extremes are tested and Matrixing design in which selected samples are tested. Bracketing is mainly used to pursue a trend initially in pre clinical studies and clinical trials. Matrixing is used to confirm a prediction of the stability information.
What are stability zones and stability conditions?
Know different climatic zones by ICH in the world for stability conditions including Temperate, Mediterranean / subtropical, Hot dry, Hot humid / tropical and hot & higher humidity zones. … According to the ICH guidelines for stability studies, the climate of the world is divided into five different zones.