What is the texture of phosphorus
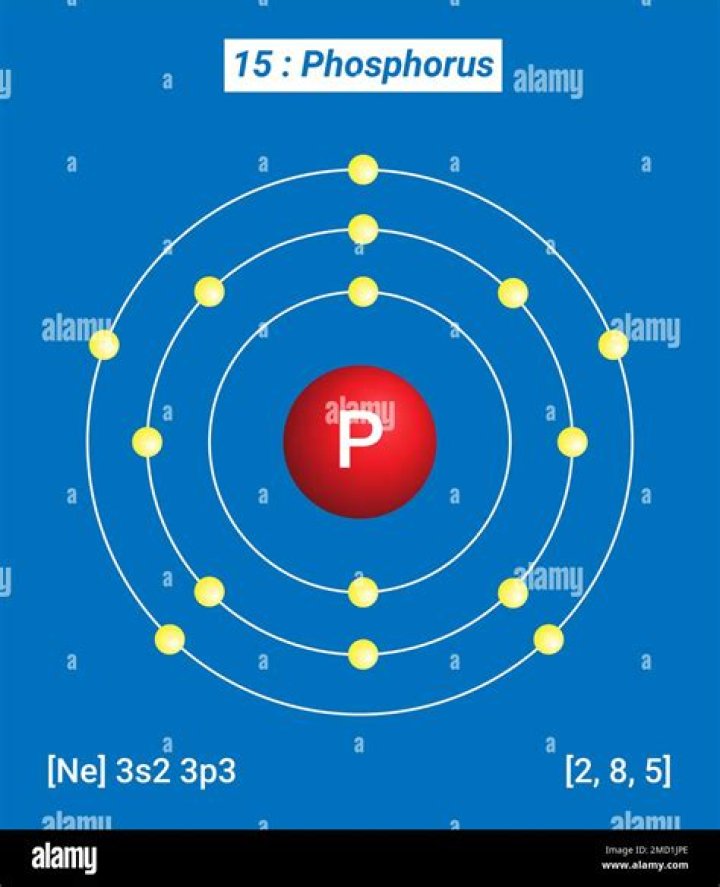
phosphorus (P), nonmetallic chemical element of the nitrogen family (Group 15 [Va] of the periodic table) that at room temperature is a colourless, semitransparent, soft, waxy solid that glows in the dark.
What does phosphorus look like?
It has a tetrahedral shape and has the formula P4. The two main forms of phosphorus are white phosphorus and red phosphorus. White phosphorus is a poisonous waxy solid and contact with skin can cause severe burns. It glows in the dark and is spontaneously flammable when exposed to air.
What colour is phosphorus?
Phosphorus in its pure form has a white colour.
What are the characteristics of phosphorus?
Characteristics: White phosphorus is a highly reactive, waxy, white-yellow, transparent solid with acrid fumes. It emits a weak green glow (luminescence) in the presence of oxygen. It is insoluble in water, but soluble in carbon disulfide.Is phosphorus a luster?
Phosphorus is a nonmetal, so it does not have a shiny luster.
Where is phosphorus element found?
Where is phosphorus found on Earth? Phosphorus is not found in its pure elemental form on Earth, but it is found in many minerals called phosphates. Most commercial phosphorus is produced by mining and heating calcium phosphate. Phosphorus is the eleventh most abundant element in the Earth’s crust.
What type of solid is white phosphorus?
White phosphorus is a colorless, white, or yellow waxy solid with a garlic-like odor. It does not occur naturally, but is manufactured from phosphate rocks. White phosphorus reacts rapidly with oxygen, easily catching fire at temperatures 10 to 15 degrees above room temperature.
What elements does phosphorus bond with?
It reacts with many metals and metalloids to form phosphides. Phosphorus atoms can bond with oxygen atoms to form ester groups. These can bond with carbon atoms, yielding a large number of organic phosphorus chemicals. These are found in many important biological processes.Why are there different colors of phosphorus?
The two main allotropes get their names from their colours: white phosphorus and red phosphorus. Both allotropes are reactive. White phosphorus is so reactive that it can spontaneously burst into flames when exposed to oxygen. The red phosphorus used in matches is slightly more stable.
Is Radium shiny or dull?A soft, shiny and silvery radioactive metal. Radium now has few uses, because it is so highly radioactive.
Article first time published onIs Silicon shiny or dull?
non-metals. Silicon has a shiny luster, but it is brittle and conducts electricity poorly. Some metalloids change their characteristics when they react with different elements.
Is phosphorus a p4 or P?
Answer: Phosphorus can form a P4 white phosphorus tetrahedron because it can form three bonds. … It can make a tetra-atomic P4 molecule by sharing valency electrons with three other P atoms to complete its octet.
Does red phosphorus glow in the dark?
White phosphorus glows in the dark while red phosphorus does not glow.
Is pencil lead shiny or dull?
Unlike most metals, lead is soft, dull, and not very good at conducting electricity. Powdered lead burns with a blue-white flame. The powdered metal is pyrophoric. Pencil lead is actually the graphite form of carbon, but lead metal is soft enough to leave a mark.
Is sulfur shiny or dull?
Object /MaterialAppearanceHardnessIronShinyVery hardCoalDullNot very hardSulphurDullNot very hardAluminiumShinyVery hard
Is sodium lustrous or dull?
If you ever get a chance to see pure sodium metal, you’ll notice that it is silvery and lustrous. Pure sodium metal is very soft and can be cut with a dull knife. It can conduct heat and electricity; however it is rarely used for this purpose.
Is yellow phosphorus an element?
Yellow phosphorus (white phosphorus) is a significantly more hazardous form of the element and may be present as a contaminant in red phosphorus.
Is phosphorus a covalent solid?
SubstanceΔHsub (kJ/mol)Average Bond Energy (kJ/mol)phosphorus (s)58.98201sulfur (s)64.22226iodine (s)62.42149
What element is white phosphorus?
White phosphorus (see images) contains four phosphorus atoms in a tetrahedral arrangement. It has an unpleasant, garliclike odor and is extremely toxic (see hazard information table). It is unstable in air—first forming white fumes before bursting into flames.
What are allotropes of phosphorus?
The main allotropes of phosphorus include the white phosphorus, red phosphorus and black phosphorus. In addition to these, there also exists a violet phosphorus.
Which allotrope of phosphorous is translucent white waxy solid?
(A) White waxy solid (A) is white phosphorus. When white phosphorus is heated in an inert atmosphere at 573K, it changes to red phosphorus.
Which Colour is not taken by phosphorus?
Red phosphorus is not soluble in water, alkalies and carbon disulphide because it exists in a polymeric structure. Under normal conditions, it remains steady. So, it does not show chemiluminescence.
Is Phosphate an element or compound?
phosphate, any of numerous chemical compounds related to phosphoric acid (H3PO4).
Why does phosphorus have different oxidation states?
Because it is more electronegative than most metals, phosphorus reacts with metals at elevated temperatures to form phosphides, in which it has an oxidation number of -3.
What type of bond does phosphorus form?
Unlike nitrogen, phosphorus has 2 vacant d-orbitals. So it can accommodate 5 electrons while forming covalent bonds. Therefore, it forms 5 covalent bonds.
Are alkali metals shiny?
The alkali metals – lithium, sodium, potassium, rubidium, caesium and francium — are all shiny, soft and highly reactive. They’re soft enough to cut with a knife, and freshly cut surfaces tarnish rapidly in the air. They have to be stored under soil, and are never found naturally as free elements.
What Colour is radium?
radium (Ra), radioactive chemical element, the heaviest of the alkaline-earth metals of Group 2 (IIa) of the periodic table. Radium is a silvery white metal that does not occur free in nature.
Is radium a solid liquid or gas?
NameRadiumStandard stateSolid at 298 KCAS Registry ID7440-14-4Group in periodic table2Group nameAlkaline earth metal
What are the shiny elements?
These metals are aluminum (Al), gallium (Ga), indium (In), tin (Sn), thallium (Tl), lead (Pb), and bismuth (Bi). These metals are solid, shiny, and good conductors of electricity and heat, all properties that we normally associate with metals.
What is the shiniest element?
Products. Iridium is the second-most dense element (after osmium) and the most corrosion resistant metal known, making it the shiniest precious metal. Iridium is alloyed with platinum to produce highly corrosion resistant electrical contacts for spark plugs.
Do metalloids have luster?
Luster: Metalloids can be either shiny or dull in their appearance. Malleability: Metalloids can be malleable and ductile, like metals. They also can be brittle, similar to nonmetals. Conductivity: Metalloids have intermediate heat and electrical conductivity.



