Where is protease naturally found
They’re also called peptidases, proteases or proteinases. In the human body, they are produced by the pancreas and stomach. While proteolytic enzymes are most commonly known for their role in the digestion of dietary protein, they perform many other critical jobs as well.
Where does protease break down in the body?
Proteins are digested in the stomach and small intestine. Protease enzymes break down proteins into amino acids. Digestion of proteins in the stomach is helped by stomach acid, which is strong hydrochloric acid. This also kills harmful microorganisms that may be in the food.
Where is glucoamylase found in the human body?
Humans and other animals produce glucoamylase produced in the mouth and pancreas, but it may also be derived from non-animal sources.
What is the source of protease?
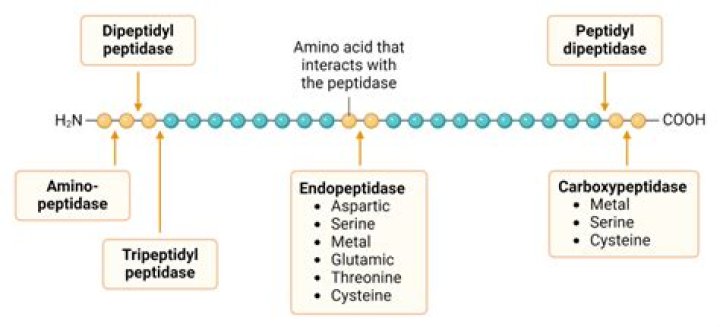
2.1 Sources of Proteases. Proteases from all sources, that is, bacteria, fungi, virus, plants, animals, and humans, have been identified because of their important physiological roles. On the basis of site of action on protein substrates, they are broadly classified as endo-peptidases or exo-peptidases.Do proteases digest themselves?
So, it is impossible for the proteolytic enzymes to digest themselves because they unable to bend themselves, or else their conformation will change and then becoming non-functional.
Where can cellulase be found?
According to recent enzyme market reports, the key areas of the industry where cellulase enzyme is increasingly being applied are healthcare, textile, pulp and paper, detergent, food, and beverages.
What is the function of proteases?
proteolytic enzyme, also called protease, proteinase, or peptidase, any of a group of enzymes that break the long chainlike molecules of proteins into shorter fragments (peptides) and eventually into their components, amino acids.
Can proteases destroy each other?
Can proteases destroy each other? Yes, and they do, because they themselves are proteins.Where are proteases used?
Proteases are used throughout an organism for various metabolic processes. Acid proteases secreted into the stomach (such as pepsin) and serine proteases present in theduodenum (trypsin and chymotrypsin) enable us to digest the protein in food. Proteases present in blood serum (thrombin, plasmin, Hageman factor, etc.)
How protease is produced in industry?Proteases are predominantly produced by microorganisms using submerged and solid-state fermentations. The bottleneck of production is found in downstream processes, which can account for 70 to 90% of the total production cost. Precipitation is used in addition to extraction using aqueous two-phase systems (ATPSs).
Article first time published onWhere is Sucrase produced in the body?
Sucrase is secreted by the tips of the villi of the epithelium in the small intestine.
What food contains glucoamylase?
a Bacterial Glucoamylases Glucoamylase from B. firmus digested potato, maize, and wheat starches yielding at 37 °C principally glucose, but small amounts of maltose, maltotriose, and maltotetraose are also formed.
What is glucoamylase in food?
Glucoamylase is one of the oldest and widely used biocatalysts in food industry. The major application of glucoamylase is the saccharification of partially processed starch/dextrin to glucose, which is an essential substrate for numerous fermentation processes and a range of food and beverage industries.
How is protease activated in the stomach?
The proteases are activated by a cascade initiated by enterokinase. These proteases catalyze further hydrolysis of the dietary proteins, resulting in a mixture consisting of about 50% free amino acids and 50% oligopeptides from two to eight amino acids in length.
Why does protease not digest the stomach?
2. Enzymes will not digest the lining of your mouth, stomach, or intestine. … Enzymes can bind to certain areas of the uncoiled protein and then chop it up. Also, the cells of our bodies and the mucus lining the gastrointestinal tract contain inhibitors that disable proteases (enzymes that break down proteins).
Who discovered protease?
William Beaumont’s pioneering research on gastric secretion has been germinal in the discovery of proteolytic enzymes and the elucidation of their chemical structure, physiological roles, and biochemical evolution.
Where are Endopeptidases produced?
An endopeptidase (pepsin) is the only protein-digesting enzyme in the stomach. Only later, in the small intestine, is the food mixed with exopeptidases.
What is cellulase an example of?
CellulaseEC no.3.2.1.4CAS no.9012-54-8DatabasesIntEnzIntEnz view
What detergent has cellulase?
BIOTOUCH® DCL Liquid cellulase preparation designed for household laundry detergents. BIOTOUCH® DCL intensifies fabrics’ colors and keeps the appearance of laundry bright and fresh. BIOTOUCH® DCL has great anti-greying and soil anti-redeposition properties.
Is cellulase an example of an enzyme?
Cellulases are a class of hydrolytic enzymes that catalyze cellulolysis or hydrolysis of the polysaccharide, cellulose, into monosaccharides such as β-glucose, or shorter polysaccharides and oligosaccharides. These enzymes are of considerable economic importance because of their multifarious uses.
What are examples of proteases?
Overview. Proteolytic enzymes (proteases) are enzymes that break down protein. These enzymes are made by animals, plants, fungi, and bacteria. Some proteolytic enzymes that may be found in supplements include bromelain, chymotrypsin, ficin, papain, serrapeptase, and trypsin.
Is trypsin a protease?
Trypsin is a serine protease that specifically cleaves at the carboxyl side of lysine and arginine residues. The selectivity of this enzyme is critical for reproducible protein digestion and mass spectrometry-based protein identification.
Is amylase a protease?
Types of enzymes Amylase breaks down starches and carbohydrates into sugars. Protease breaks down proteins into amino acids.
Can chymotrypsin digest itself?
Description. Chymotrypsin is a digestive enzyme that breaks down proteins (i.e., it is a proteolytic enzyme; it can also be referred to as a protease). It is naturally produced by the pancreas in the human body. … The enzymes are secreted in an inactive form (as proenzymes) so that they will not digest the pancreas.
Is sucrase found in plants?
Sucrose is the end product of photosynthesis and the primary sugar transported in the phloem of most plants. … Plant SuSy isozymes are mainly located in the cytosol or adjacent to plasma membrane, but some SuSy proteins are found in the cell wall, vacuoles, and mitochondria.
Where is sucrase found in the human body quizlet?
Where is sucrase found in the human body? On the microvilli of the small intestine. Sucrase uses ____ to cleave sucrose into two monosaccharides.
Is sucrase found in saliva?
Sucrase activity was expressed as nanomoles of glucose or fructose liberated per minute and per milliliter of saliva. According to our experience values equal to 10 nmol/min/ml or higher indicate a high sucrase activity [Karjalainen et al., 1987].
What is glucoamylase good for?
Glucoamylase is widely used in the food industry to produce high glucose syrup, and also in fermentation processes for production beer and ethanol.
Where is glucoamylase located and what does it breakdown?
Tissue distribution. Maltase-glucoamylase is a membrane-bound enzyme located in the intestinal walls. This lining of the intestine forms what is called a brush-border in which food has to pass in order for the intestines to absorb the food.
What is glucoamylase deficiency?
Disease definition. A rare intestinal disease characterized by impaired absorption of starch and short polymers of glucose due to primary small intestinal glucoamylase deficiency. Patients present in infancy or early childhood with chronic diarrhea, abdominal distention, and bloating.
How is Pullulanase produced?
It is produced as an extracellular, cell surface-anchored lipoprotein by Gram-negative bacteria of the genus Klebsiella. Type I pullulanases specifically attack α-1,6 linkages, while type II pullulanases are also able to hydrolyse α-1,4 linkages. It is also produced by some other bacteria and archaea.



