Which compound will distill first
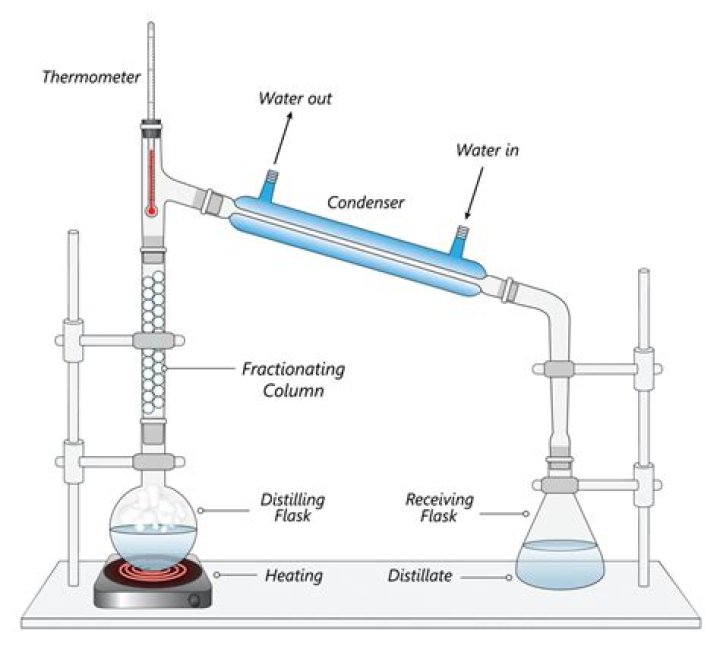
1. What determines which compound comes off first in a distillation? The boiling point determines which compound comes off first. More volatile compounds will have lower boiling points and will come off first.
What is the mixture of acetone and water?
When acetone is mixed with water it completely dissolves in it. In this type of reaction acetone is usually the solute and water is the solvent. When these two compounds are mixed there is a formation of hydrogen bonds resulting in a homogeneous solution.
How do you distill acetone?
Acetone can be distilled from water while keeping a close eye on the temperature of the solution. Best possible method is fractional distillation at controlled temperature . set the distillation system temperature to boiling point of acetone then it willbe carried over which can be cooled later.
How will you separate acetone and water by simple distillation?
Difference in the boiling points of acetone and water is 25K. The mixture of water and acetone is taken in a distillation flask and is slowly heated till acetone evaporates. Water is collected in the distillation flask and acteone vapours are condensed to get acetone. Thus the mixture is separated.What is the first step in the process of distillation?
The process of distillation begins with heating a liquid to boiling point. The liquid evaporates, forming a vapor. The vapor is then cooled, usually by passing it through pipes or tubes at a lower temperature. The cooled vapor then condenses, forming a distillate.
Why do the two components separate acetone and water?
“1) While start heating, acetone separates from the water because of the lower boiling point compared to the water. … 3) The boiling point of acetone is 56-degree Celsius. 4) The difference in boiling water was the reason for separation.
Do compounds with lower boiling points distill first?
The temperature drops because the lower-boiling compound finishes distilling before vapors of the higher-boiling compound can fill the distillation head, which then cause the head temperature to rise.
Which method will you use to separate components of air and acetone and water?
Distillation is used for the separation of components of a mixture containing two miscible liquids that have a large difference in their boiling points. The liquid with lower boiling point evaporates at first and the vapours of it can be collected at the nozzle.What kind of mixture is the acetone?
Acetone, or propanone, is an organic compound with the formula (CH3)2CO.
How do you separate acetone from alcohol and water?Fractional distillation is a method for separating a liquid from a mixture of two or more liquids. For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points.
Article first time published onWhich technique is used to separate acetone and benzene by the following method?
A mixture of acetone and benzene can be separated by fractional distillation.
How is acetone extracted from water?
There is no way that these mixtures (acetone:water, ethanol:water and methanol:water) can be separated into two layers. The best way to solve this problem (may be partially or fully) is to add a third solvent such as dichloromethane (DCM) to the the mixtures.
Does acetone react with water?
Acetone and water are freely miscible and one dissolves in the other. To a very small extent (about 0.1%), acetone also reacts with water to form the hydrate or geminal diol. Acetone and water are freely miscible and one dissolves in the other.
What is the simple distillation?
Definition: A method of separating mixtures based on differences in their volatilities in a boiling liquid mixture. The components in a sample mixture are vaporised by the application of heat and then immediately cooled by the action of cold water in a condenser.
How could you separate a mixture of acetone and naphthol?
How could you separate a mixture of acetone and alpha-naphthol? In cyclohexane alpha-naphthol is insoluble and acetone is soluble. Alpha-naphthol can be removed by filtering it from the cyclohexane acetone mixture.
What is the first method that you should apply to separate salt from water in seawater?
For example, water can be separated from salt solution by simple distillation. This method works because water has a much lower boiling point than salt. When the solution is heated, the water evaporates.
What distills first in fractional distillation?
Use a heating mantle/block or oil bath to heat the organic mixture to the appropriate temperature. You should start by setting the temperature to distill the lowest-boiling compound first, to aid in separation. Note the ring of condensate rising slowly up the fractionating column.
What are the 5 steps of distillation?
- Distillation process to separate ethanol from water. …
- The ethanol evaporates first, cools, then condenses.
- The water left evaporates, cools, then condenses.
Why is an ice bath used in distillation?
Since the vapor pressure of water at 20 °C is roughly 2.3 kPa, then 2.3/101.325 or 0.023 would be the fraction of water that would not condense and would pass out of the condenser. This is why the distillate is frequently chilled in an ice bath during the distillation.
What mixtures can be separated by distillation?
Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or in the separation of two or more liquids having different boiling points, as in the separation of gasoline, kerosene, and lubricating oil from crude oil.
What is the boiling point of acetone?
Acetone is a colourless liquid with mildly pungent, somewhat aromatic odour. The melting point of acetone is -94.7°C (range: -94.6°C to – 95.6°C), the boiling point is 56.05°C (range: 56.05 to – 56.5°C).
How can two miscible liquids like acetone and water be separated Name the process explain with a diagram?
Fractional distillation is the process of separating two (or more) miscible liquids by distillation, the distillate being collected in fractions boiling at different temperatures. A mixture of two miscible liquids can be separated by the process of fractional distillation.
Is acetone soluble in water?
Acetone is a manufactured chemical that is also found naturally in the environment. It is a colorless liquid with a distinct smell and taste. It evaporates easily, is flammable, and dissolves in water. It is also called dimethyl ketone, 2-propanone, and beta-ketopropane.
What is acetone derived?
Acetone is manufactured from the basic raw materials of benzene and propylene. These materials are first used to produce cumene, which is then oxidised to become cumene hydroperoxide, before being split into phenol and its co-product, acetone.
What are the elements in acetone?
Acetone is a naturally occurring compound also known as propanone. Composed of the elements carbon, hydrogen, and oxygen, acetone presents as a clear liquid that is highly flammable and often used as cleaner in industrial settings.
Which method is used to separate air water?
An air separation plant separates atmospheric air into its primary components, typically nitrogen and oxygen, and sometimes also argon and other rare inert gases. The most common method for air separation is fractional distillation.
Why Cannot we separate a mixture of acetone and water using a separating funnel?
(b) Acetone and water mixture cannot be separated by using a separating funnel because they are miscible liquids. They are rather separated by fractional distillation.
Which technique is used to separate the components of air?
Fractional distillation is the technique that is used for obtaining different components from the air.
Why is condenser used in distillation process?
In the process of distillation, a liquid is converted into its vapor and the vapor is then condensed back to liquid on cooling. The condenser has a jacket with two outlets through which water is circulated. Hence, to provide efficient cooling, a condenser is used.
Which technique is widely used in industry to separate components of mixture and also to purify them?
Answer: Chromatography is the process to separate the components as well as to purify them.
Which of the following method can be used to separate two compounds with different solubility?
Fractional crystallization is used to separate two compounds with different solubilities in a solvent.