Which elements have the same number of protons and neutrons
Chemical SymbolNamePbLeadPlumbum
What element has the same proton neutron and electron?
Elementary ParticleChargeMassProton+11Neutron01Electron−1~0
Are there always the same number of protons and neutrons?
The number of protons is always the same in atoms of the same element. The number of neutrons can be different, even in atoms of the same element. Atoms of the same element that contain the same number of protons, but different numbers of neutrons, are known as isotopes.
Do all atoms have the same number of protons neutrons and electrons?
Atoms always have an equal number of protons and electrons, and the number of protons and neutrons is usually the same as well. Adding a proton to an atom makes a new element, while adding a neutron makes an isotope, or heavier version, of that atom.Can two different elements contain the same number of protons or neutrons?
Atoms from two different elements may have the same number of neutrons, but never the same number of protons. The number of protons is unique to the element and represents the atomic number.
Do elements always have the same number of protons and electrons?
Explanation: Number of protons always equal number of electron, but that’s the case with a neutral atom. A neutral atom is an atom of any element that has no electric charge over it. … Thus, in an ionized atom or simply an ion, the number of protons and electrons are not the same but in a neutral atom they are.
Which chemical elements have the same number of protons?
NameHydrogenNeutrons0Electrons1Atomic Number (Z)11
What types of atoms have the same number of electrons and protons?
Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element’s atomic number (Z) and distinguishes one element from another.Does every atom of the same element have the same atomic number?
Each element has its own atomic number, which is equal to the number of protons in its nucleus. Isotopes of an element contain different numbers of neutrons. Elements are represented by an atomic symbol.
Why do atoms have equal numbers of protons and electrons?Actually the proton and electron count of an atom are equal only when the atom is neutral in charge. … The electrons are found in orbitals surrounding the nucleus. In order for the atom to remain electrically neutral the protons and electrons must balance each other.
Article first time published onDo all atoms of the same element have the same number of protons?
All atoms of an element have the same number of protons, and every element has a different number of protons in its atoms. … Atoms are neutral in electrical charge because they have the same number of negative electrons as positive protons (Table 2.4. 1).
Are all protons the same?
All protons are identical. For example, hydrogen protons are exactly the same as protons of helium and all other elements, or pure substances. However, atoms of different elements have different numbers of protons. … The number of protons in an atom determines the electrical charge of the nucleus.
Can 2 elements have the same atomic number?
No, because each element has its own specific property, BUT certain elements have similar properties, like elements in the same family. … Meaning each element has more protons in their atoms than the previous.
How can two different elements have the same mass number?
Atoms of an element with the same mass number make up an isotope of the element. Different isotopes of the same element cannot have the same mass number, but isotopes of different elements often do have the same mass number, e.g., carbon-14 (6 protons and 8 neutrons) and nitrogen-14 (7 protons and 7 neutrons).
Why can atoms of two different element have the same mass number but not the same atomic number?
Atoms of the same chemical element do not always have the same mass because, although the number of protons in the nucleus is the same for all atoms of the same element, the number of neutrons is not.
What do elements in the same group have in common?
The elements in each group have the same number of valence electrons. As a result, elements in the same group often display similar properties and reactivity.
When the proton number and electron number are unequal?
is an ion When the proton number and electron number are unequal, the atom or molecule has a net positive (cation) or negative charge (anion). 11 The number of electrons will equal the number of protons in an electrically neutral atom.
How are atoms of the same element similar?
Atoms of the same element consist of the same atomic number that is number of protons or electrons and different mass number that is number of neutrons. This variation or change in the number of neutrons in an atom gives rise to different isotopes of an element. An element with different isotopes occurs in nature.
What are atoms of the same element with the same number of protons and electrons but different number of neutrons?
Isotopes are atoms of the same element that have different numbers of neutrons but the same number of protons and electrons. The difference in the number of neutrons between the various isotopes of an element means that the various isotopes have different masses.
What are 2 atoms with the same number of protons and electrons but different number of neutrons called?
Two atoms with the same atomic number, but different mass numbers (same number of protons, different number of neutrons), are called isotopes, or isotopic nuclides. Having different numbers of neutrons changes the mass of these atoms, so isotopes have slight variations in their physical and chemical behavior.
What has the same number of protons but in the number of neutrons?
Atoms of the same element, containing the same number of protons, but different numbers of neutrons, are known as isotopes. Isotopes of any given element all contain the same number of protons, so they have the same atomic number (for example, the atomic number of helium is always 2).
Does the atomic number equal the number of protons?
The number of protons in the nucleus of the atom is equal to the atomic number (Z). The number of electrons in a neutral atom is equal to the number of protons.
Which two atoms have the same number of neutrons?
Isotones and isobars have great significance for studying of nuclide stability. Two nuclides (a type of a nucleus with some values of Z and N) that have the same number of neutrons N have the same number of neutrons.
What are protons and neutrons made of?
Protons and neutrons are composed of two types: up quarks and down quarks. Each up quark has a charge of +2/3. Each down quark has a charge of -1/3. The sum of the charges of quarks that make up a nuclear particle determines its electrical charge.
What are protons and neutrons?
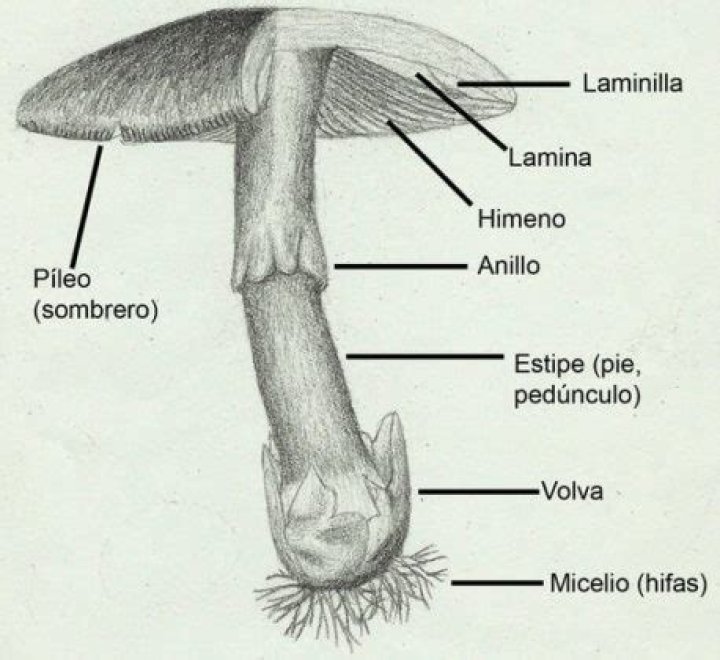
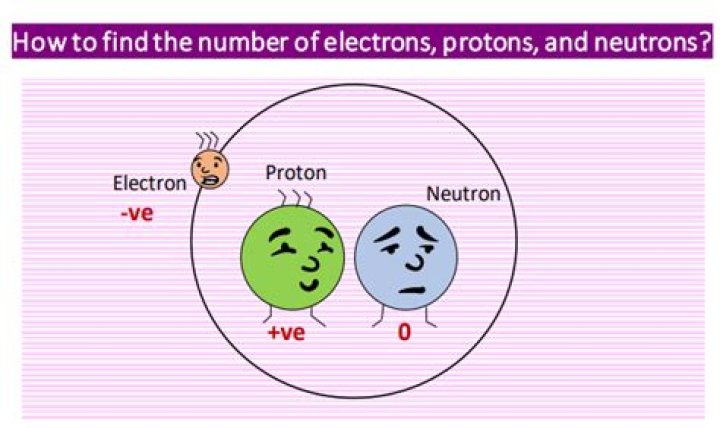
Atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons and neutrons are in the center of the atom, making up the nucleus. Electrons surround the nucleus. Protons have a positive charge. … Neutrons have no charge.
Why do elements have the same atomic number?
The atomic number is the same as the number of protons. … The atomic mass is the sum of the number of protons and neutrons. For ions (which may either have a positive or negative charge), only the number of electrons are being changed. If the ion is negative it means that an electron has been added to the configuration.
Which element has the same atomic number?
Isotopes are atoms that have the same atomic number (and are therefore of the same element) but different mass numbers. The composition of atoms of the naturally occurring isotopes of carbon are shown in Table 4.2. we can write carbon-12, carbon-13, and carbon-14.
Do all protons have the same mass?
To answer your specific question: protons are stable, so they all have the same mass.
What do protons and neutrons have in common How are they different?
Protons and neutrons have almost the same mass and are both located in the nucleus of the atom. Protons are charged particles. Neutrons are neutral particles. … The positive charge of the protons in the nucleus is balanced by the negative charge of the electrons.