Which functional groups are basic
Charged (forms R-NH 3+start subscript, 3, end subscript, start superscript, plus, end superscript) at the pH of most biological systems. Since amino groups can remove H +start superscript, plus, end superscript from solution, they are considered basic.
Which functional groups are bases?
Amino groups can also act as bases, which means that the nitrogen atom can bond to a third hydrogen atom as shown in the image below. Once this occurs, the nitrogen atom gains a positive charge and can now participate in ionic bonds. The amine functional group can exist in a deprotonated or protonated state.
Are hydroxyl groups Basic?
Non-acidic Functional Groups The hydroxyl, which is simply an OH attached to the molecule, could conceivably lose it’s hydrogen, making it acidic, but that is not how the molecule normally interacts.
What makes a functional group Basic?
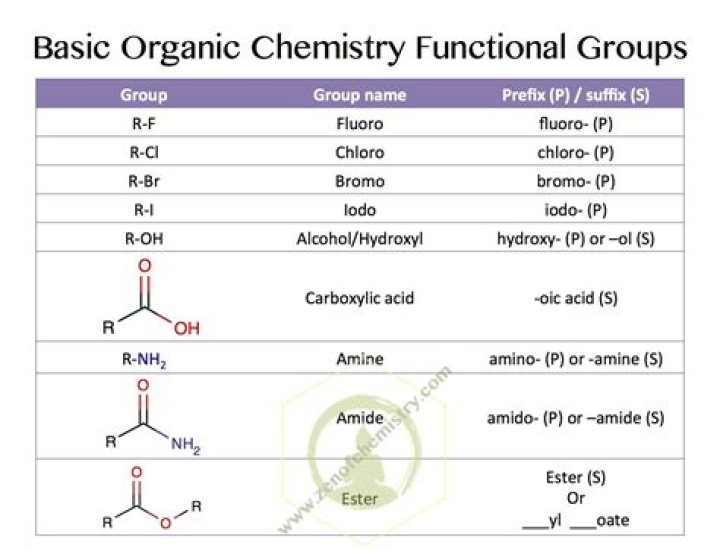
Classifying Functional Groups Functional groups are usually classified as hydrophobic or hydrophilic depending on their charge or polarity. An example of a hydrophobic group is the non-polar methane molecule. … Charged, accepts H+ to form NH3+. Since amino groups can remove H+ from solution, they are considered basic.Are amine functional groups Basic?
Compounds containing a nitrogen atom bonded in a hydrocarbon framework are classified as amines. Compounds that have a nitrogen atom bonded to one side of a carbonyl group are classified as amides. Amines are a basic functional group. Amines and carboxylic acids can combine in a condensation reaction to form amides.
Is phosphate acidic or basic?
Phosphate ion is a reasonably strong base. It hydrolyzes in water to form a basic solution.
Are amines Basic?
Because amines are basic, they neutralize acids to form the corresponding ammonium salts R3NH+. When formed from carboxylic acids and primary and secondary amines, these salts thermally dehydrate to form the corresponding amides.
Is methyl group a functional group?
The methyl group is the only nonpolar functional group in our class list above. The methyl group consists of a carbon atom bound to three hydrogen atoms. … This means that methyl groups are unable to form hydrogen bonds and will not interact with polar compounds such as water.Is methyl acidic or basic?
Functional GroupPropertiesMethylNonpolarCarbonylPolarCarboxylCharged (forms R-COO −start superscript, minus, end superscript) at the pH of most biological systems. Since carboxyl groups can release H +start superscript, plus, end superscript into solution, they are considered acidic.
How do you know if a functional group is acidic or basic?The functional group helps to determine whether something is acid, low pH, or basic and has a high pH. An example of an acidic functional group is a carboxyl. The carboxyl functional group is acidic because it is a proton (H+) donator when in solution.
Article first time published onAre carbonyl groups Basic?
Reactions with acids: – The carbonyl oxygen is weakly basic. – Both Bronsted and Lewis acids can interact with a lone pair of electrons on the carbonyl oxygen. Addition Reactions – Carbonyl groups in aldehydes and ketones undergo addition reactions. – This is one of the most important reactions of the carbonyl group.
Is a carbonyl group polar?
Thus, molecules containing the carbonyl group are polar. Compounds containing a carbonyl group have higher melting and boiling points than hydrocarbons containing the same number of carbon atoms and are more soluble in polar solvents such as water.
Where are carbonyl groups found?
The carbonyl group is found in many other functional groups, from aldehydes and anhydrides to ketones and carboxylic acids, just to name a few. The key point to remember is that whenever you see the formula carbon-oxygen double bond (C=O), you know a carbonyl is present in that functional group.
Are amides basic?
Compared to amines, amides are very weak bases. … Therefore, amides don’t have as clearly noticeable acid–base properties in water. This relative lack of basicity is explained by the withdrawing of electrons from the amine by the carbonyl.
Is thiol a functional group?
In organic chemistry, a thiol is a compound that contains the –SH functional group, which is the sulfur analog of a hydroxyl or alcohol group. The functional group is referred to as either a thiol group or a sulfhydryl group. Thiols are more traditionally referred to as mercaptans.
Is ch2oh a functional group?
There is CH2 is present with OH therefore in CH2 one double bond is present between one hydrogen and one carbon So, it is present from alkene group and there is one carbon So methene is used and alcohol is functional group .
Is aniline basic?
Basicity. Aniline is a weak base. Aromatic amines such as aniline are, in general, much weaker bases than aliphatic amines.
Which is more basic acetamide or aniline?
Explanation :- aniline is more basic than acetamide because in acetamide, lone pair of nitrogen is delocalised to more electronegative element oxygen. In Aniline lone pair of nitrogen delocalised over benzene ring.
Is ammonium more basic than ammonia?
Study Notes. The lone pair of electrons on the nitrogen atom of amines makes these compounds not only basic, but also good nucleophiles. … The ammonium ions of most simple aliphatic amines have a pKa of about 10 or 11. However, these simple amines are all more basic (i.e., have a higher pKa) than ammonia.
Is NH3 acid or base?
Ammonia, NH3, is a Lewis base and has a lone pair. It will donate electrons to compounds that will accept them. Donation of ammonia to an electron acceptor, or Lewis acid. There may be anionic or neutral Lewis bases.
Is CO3 acidic or basic?
Carbonates (CO3–) are actually weak bases. Carbonic Acid (H2CO3) breaks apart to form H+ and HCO3- or to make two H+’s and CO3—.
Does phosphorus react with bases?
These halides are used to synthesize organic phosphorus chemicals. Phosphorus reacts with sulfur to form several sulfides that are used in the manufacture of organic chemicals and matches. It reacts with many metals and metalloids to form phosphides. Phosphorus atoms can bond with oxygen atoms to form ester groups.
Is amino group acidic or basic?
Amino AcidClassificationpIglutamic acidnegatively charged (acidic)3.2
Is amino group hydrophobic or hydrophilic?
Hydrophilic functional groups include hydroxyl groups (resulting in alcohols though also found in sugars, etc.), carbonyl groups (giving rise to aldehydes and ketones), carboxyl groups (resulting in carboxylic acids), amino groups (i.e., as found in amino acids), sulfhydryl groups (giving rise to thiols, i.e., as found …
Is Bromothymol Blue an acid or base?
Bromothymol blue is an acidic pH chromoionophore where acid to base change corresponds to its neutral to anionic form, respectively. For this reason it can be electrostatically immobilized on the surface on anion-exchanger resin like Amberlite IRA 401. Solutions of different concentration of the indicator were used.
Is alkene a functional group?
NumberPrefix7hepta
Is ethyl a functional group?
Chemical classEsterGroupEsterFormulaRCO2R’Prefix/ExampleEthyl acetate (Ethyl ethanoate)
Is NH a functional group?
In organic chemistry, the most common functional groups are carbonyls (C=O), alcohols (-OH), carboxylic acids (CO2H), esters (CO2R), and amines (NH2). It is important to be able to recognize the functional groups and the physical and chemical properties that they afford compounds.
How do you know when something is acidic or basic?
If the pH is lower than 7, the solution is acidic. When pH is higher than 7, the solution is basic. These numbers describe the concentration of hydrogen ions in the solution and increase on a negative logarithmic scale.
Is alcohol functional group acidic or basic?
By the Arrhenius definition of an acid and base, alcohol is neither acidic nor basic when dissolved in water, as it neither produces H+ nor OH- in solution. They are generally weak acids. Alcohols are very weak Brønsted acids with pKa values generally in the range of 15 – 20.
Is ether a carbonyl group?
Ethers are compounds with an oxygen atom bonded to two alkyl groups. Aldehydes and ketones contain the carbonyl functional group. In an aldehyde, the carbonyl is at the end of a carbon chain, while in a ketone, it is in the middle.



