Which type of bond is formed when electrons are transferred from one atom to another
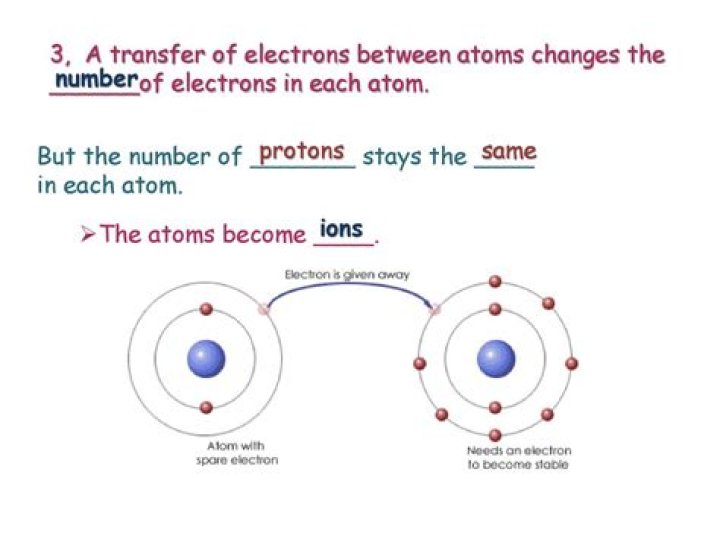
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
Which type of bond is formed by the transfer of electrons from one atom to another quizlet?
An ionic bond is a bond that forms when electrons are transferred from one atom to another. During ionic bonding, the atoms become oppositely charged ions.
Why do nonmetals form covalent bonds?
Covalent bonding occurs when pairs of electrons are shared by atoms. … Nonmetals will readily form covalent bonds with other nonmetals in order to obtain stability, and can form anywhere between one to three covalent bonds with other nonmetals depending on how many valence electrons they posses.
When electrons are transferred from one atom to another both atoms become?
Ionic bonds form when an electron of one atom is transferred to another atom. The two atoms become oppositely charged ions and attract each other.What is ionic bond and covalent bonds?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
Do metalloids form covalent bonds?
Metalloids and non-metals not only form covalent bonds by sharing, but can form ionic bonds either by losing or gaining electrons.
What happens when electrons transfer?
ionic bonding … stems from the transfer of electrons from one atom to another. When such a transfer occurs, all the valence electrons on the more electropositive element (from one of the first three groups on the left in the periodic table) are removed to expose the core of the atom.
What type of atoms form covalent bonds?
In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals. For example, in water (H2O) each hydrogen (H) and oxygen (O) share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom.What is single covalent bond?
A single covalent bond is when only one pair of electrons is shared between atoms. A sigma bond is the strongest type of covalent bond, in which the atomic orbitals directly overlap between the nuclei of two atoms.
What type of electrons are available to form bonds?valence electrons: The electrons of an atom that can participate in the formation of chemical bonds with other atoms. They are the furthest electrons from the nucleus.
Article first time published onHow do the covalent bonds form between atoms?
A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions. Shared electrons located in the space between the two nuclei are called bonding electrons. The bonded pair is the “glue” that holds the atoms together in molecular units.
What kind of particle is formed after covalent bonding?
Particles formed from the covalent bonding of atoms are called ions. Polar molecules share their electrons equally.
Why are electrons transferred in ionic bonds?
It is the power of an atom in a molecule to attract electrons to itself. When the electronegativity difference between the two atoms is greater than 2.0, the bond is considered ionic; i.e. there is complete transfer (between the two atoms) of the electrons in the bond.
What causes electron transfer?
Electron transfer results from a combination of oxidation and reduction. A species cannot be oxidized unless another species accepts the electrons and is reduced. That is, oxidation causes reduction and vice versa.
Does antimony form covalent bonds?
The electronic structure of antimony closely resembles that of arsenic, with three half-filled orbitals in the outermost shell. Thus it can form three covalent bonds and exhibit +3 and −3 oxidation states.
Does Silicon form covalent bonds?
Overview. Each silicon atom has four valence electrons which are shared, forming covalent bonds with the four surrounding Si atoms.
Do metalloids gain or lose electrons when bonding?
Metalloids – can either lose or share electrons when they combine with other elements.
What are 3 types of covalent bonds?
- Single bonds occur when two electrons are shared and are composed of one sigma bond between the two atoms.
- Double bonds occur when four electrons are shared between the two atoms and consist of one sigma bond and one pi bond.
How are ionic bonds formed?
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
What are the 4 types of covalent bonds?
- Single Covalent Bond.
- Double Covalent Bond.
- Triple Covalent Bond.
Which pairs would form covalent bonds?
Oxygen and hydrogen share electrons with each other. … The atoms of propane will form covalent bonds because carbon and hydrogen are both nonmetals.
Which atoms will bond when valence electrons are transferred from one atom to the other?
Ionic bonding Definition: An ionic bond is formed when valence electrons are transferred from one atom to the other to complete the outer electron shell. Example: A typical ionically bonded material is NaCl (Salt): The sodium (Na) atom gives up its valence electron to complete the outer shell of the chlorine (Cl) atom.
What type of atom forms when an atom loses electrons?
An atom loses electrons to form a cation , that is a positively charged ion (and one that is attracted towards the negatively charged terminal, the cathode ). Both charge and mass have been conserved.
What happens when atoms bond?
A chemical bond is formed when atoms are held together by attractive forces. This attraction occurs when electrons are shared between atoms, or when electrons are exchanged between the atoms that are involved in the bond. … In other words one atom gains an electron while the other loses an electron.
How can you tell which bond is formed?
One way to predict the type of bond that forms between two elements is to compare the electronegativities of the elements. In general, large differences in electronegativity result in ionic bonds, while smaller differences result in covalent bonds.
Does an electric charge form on atoms when a covalent bond is formed between them?
In a covalent bond, the stability of the bond comes from the shared electrostatic attraction between the two positively charged atomic nuclei and the shared, negatively charged electrons between them. … When atoms combine by forming covalent bonds, the resulting collection of atoms is called a molecule.
What kind of chemical bond is formed when electrons are gained and lost from atoms?
Formation of ionic bond An ionic bond can be formed after two or more atoms loss or gain electrons to form an ion. Ionic bonds occur between metals, losing electrons, and nonmetals, gaining electrons. Ions with opposite charges will attract one another creating an ionic bond.
What kind of particle is produced after complete transfer of electrons?
Ionic bonding is the complete transfer of valence electron(s) between atoms. It is a type of chemical bond that generates two oppositely charged ions. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion.
When an atom loses one electron the resulting particle is called?
If an atom loses or gains electrons, it will become a positively or negatively charged particle, called an ion. The loss of one or more electrons results in more protons than electrons and an overall positively charged ion, called a cation.
What is formed after an atom gains electrons?
Forming an Ion An atom that loses one or more valence electrons to become a positively charged ion is known as a cation, while an atom that gains electrons and becomes negatively charged is known as an anion.
What are the 3 types of electron transfer?
- Inner-sphere electron transfer.
- Outer-sphere electron transfer.
- Heterogeneous electron transfer.