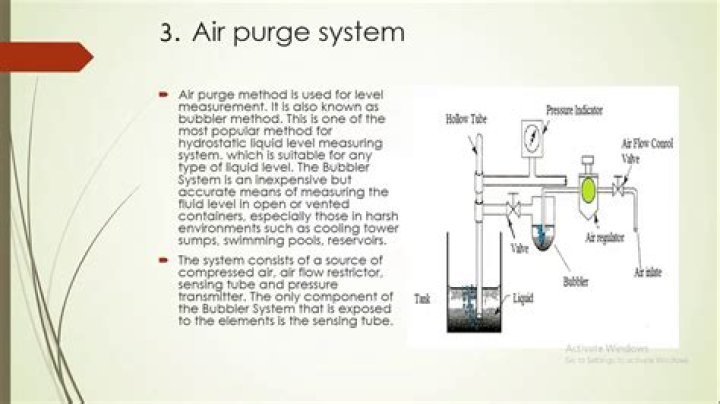
Why is Equatorial more stable

Substituents prefer equatorial rather than axial positions in order to minimize the steric strain created of 1,3-diaxial interactions. The more stable conformation will place the larger substituent in the equatorial position.
Why equatorial conformation of methylcyclohexane is more stable than corresponding axial conformation?
When the methyl group in the structure above occupies an axial position it suffers steric crowding by the two axial hydrogens located on the same side of the ring. The conformation in which the methyl group is equatorial is more stable, and thus the equilibrium lies in this direction.
Which conformation of methylcyclohexane is more stable?
The most stable conformation of methylcyclohexane is the chair conformation in which the methyl group is equatorial. The alternative chair conformation, in which the methyl group is axial, is 7.3 kJ/mol higher in energy.
Why is Equatorial cyclohexane more stable?
Monosubstituted Cyclohexanes In the previous section, it was stated that the conformation in which the methyl group is equatorial is more stable because it minimizes steric repulsion, and thus the equilibrium favors the more stable conformer.Why axial position is less stable than Equatorial?
Because axial bonds are parallel to each other, substituents larger than hydrogen generally suffer greater steric crowding when they are oriented axial rather than equatorial. Consequently, substituted cyclohexanes will preferentially adopt conformations in which the larger substituents assume equatorial orientation.
Why is methylcyclohexane more stable than axial methylcyclohexane?
Because this process is rapid at room temperature, methylcyclohexane is a mixture of two conformational diastere- omers (Sec. 6.10A). Because diastereomers have different energies, one form is more stable than the other. Equatorial methylcyclohexane is more stable than axial methylcyclohexane.
Which is more stable of the axial and equatorial bonds?
Axial and equatorial are types of bonds found in the ‘chair’ conformation of cyclohexane. The chair conformation of cyclohexane has to lowest totally energy and is, therefore, the most stable.
Why is the energy difference for the tert Butylcyclohexane so much larger than the methylcyclohexane?
For example, the difference in energy between the two chair conformations of tert-butyl cyclohexane (24 kJ/mol) is much larger than for methylcyclohexane (7 kJ/mol), because a tert-butyl group is larger than a methyl group and results in more energetically unfavorable 1,3-diaxial interactions.Is methylcyclohexane more stable than cyclohexane?
METHYLCYCLOHEXANE CONFORMATIONS Equatorial methyl cyclohexane is the more stable conformation. When the ring flip occurs, however, it converts to axial methylcyclohexane. These two conformations are in rapid equilibrium at room temperature, but can be frozen out as distinct compounds at -78degrees.
Which cyclohexane is more stable?The most stable conformation of cyclohexane is the chair form shown to the right. The C-C-C bonds are very close to 109.5o, so it is almost free of angle strain. It is also a fully staggered conformation and so is free of torsional strain.
Article first time published onHow do you know which cyclohexane is more stable?
To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
Which conformation of cyclohexane is highly unstable?
The boat conformation of cyclohexane is not a very stable form due to the torsional strain applied to the cyclohexane molecule. The stability of this form is further affected by steric interactions between the hydrogen atoms.
Is methylcyclohexane more stable than t butyl cyclohexane?
Solving for the equilibrium constant K shows that the equatorial is preferred about 460:1 over axial. This means that 1-tert-butyl-1-methylcyclohexane will spend the majority of its time in the more stable conformation, with the tert-butyl group in the equatorial position.
In which dimethyl cyclohexane both the methyl groups are on equatorial position in its most stable chair confirmation?
The more stable chair conformation of trans-1,2-dimethylcyclohexane has the two methyl groups in the equatorial position.
How do you determine the most stable conformation?
To find the most stable conformation, we choose the form with the least number of large axial groups; the least stable will have the most number of axial groups.
Does axial or equatorial react faster?
The axial sulfonyl chloride 6 was found to react 71 times faster than the equatorial epimer 8 with triethylamine in the presence of aniline at -25″, a reaction which evidently proceeds via the sulfene 7.
Why are axial positions stable?
When the smaller group (methyl) is axial, the space above the cyclohexane ring is less crowded and so this conformation is energetically more favourable.
Why are axial bonds longer than equatorial?
The axial bonds are longer than equatorial bonds because of greater repulsion from equatorial bonds. The 2-axial bonds are at 90º degree angle to 3-equatorial bonds while all equatorial bond are at 120º angle to each other.
Is the methyl group on your methylcyclohexane in an axial or an equatorial position?
Take a look at the new conformation of methylcyclohexane, with the methyl group in an equatorial position.
What's the difference between axial and equatorial position?
The key difference between axial and equatorial position is that axial bonds are vertical while equatorial bonds are horizontal. … A conformation is a shape a molecule can take due to the rotation around one or more of its bonds.
What is equatorial position in chemistry?
Equatorial: In cyclohexane, a bond which is perpendicular to the axis of the ring (i.e., the bond lies along the equator of the chair), or a group attached by such a bond.
What determines the preference for axial or equatorial position for the oxygen atom in this compound?
The formal charge determines the preference for whether the oxygen is in axial or equatorial position for this compound. The oxygen and Sulfur on the axial positioned molecule each have FCs of 0 so it is the preferred orientation.
Which is the most stable structure of 1 isopropyl 4 methylcyclohexane structure?
Answer: Here, in case of 1-chloro-4-methylcyclohexane, CH3 and Cl are when placed at equatorial position in chair form of cyclohexane, is most stable.
Which Stereoisomer is more stable?
Trans isomers are more stable as in trans isomers same group or atom on two different carbon atoms are located on the opposite sides of the carbon-carbon double bond and hence the trans molecule is non-polar. The polarity on both the sides cancels out. So, trans-isomer stability is more than cis.
Which of the following ring system is more stable?
Cyclohexane is the most stable Cycloalkane.
How is methylcyclohexane made?
It can be also produced by hydrogenation of toluene: CH3C6H5 + 3 H2 → CH3C6H. Methylcyclohexane, as a component of a mixture, is usually dehydrogenated to toluene, which increases the octane rating of gasoline.
Why is there an energy difference between axial and equatorial methylcyclohexane?
The “Equatorial” Methyl Conformation Encounters Fewer Gauche Interactions Than the Axial Methyl Conformation. … This gauche interaction is an example of van der Waals strain, which is what makes the axial conformer higher in energy. There is actually a second gauche interaction if you look along C-1 to C-6 .
Which chair conformation has the highest energy?
The chair conformation has the lowest energy, as it has the lowest values for angular and torsional strain (they are both equal to zero). The half-chair conformation has the highest energy – due to high angular strain and eclipsed conformation for hydrogens in the flipping part of the molecule (high torsional strain).
How do you know which chair conformation has the highest energy?
The higher energy chair conformation contains one axial methyl group and one equatorial methyl group. The lower energy chair conformation contains one axial methyl group and one equatorial methyl group. The higher energy chair conformation contains two axial methyl groups.
Why is cyclohexane more stable than hexane?
This is due to the fact that cyclohexane has 2 hydrogens less compared to hexane. The volume cyclohexane occupies is lower than hexane due to its cyclic structure. Hence, there’s a higher number of cyclohexane molecules in a given molecule when compared with hexane.
Which is the least stable conformation of cyclohexane?
The least stable conformation of cyclohexane is half chair conformation.