Why LDPE has a low melting point
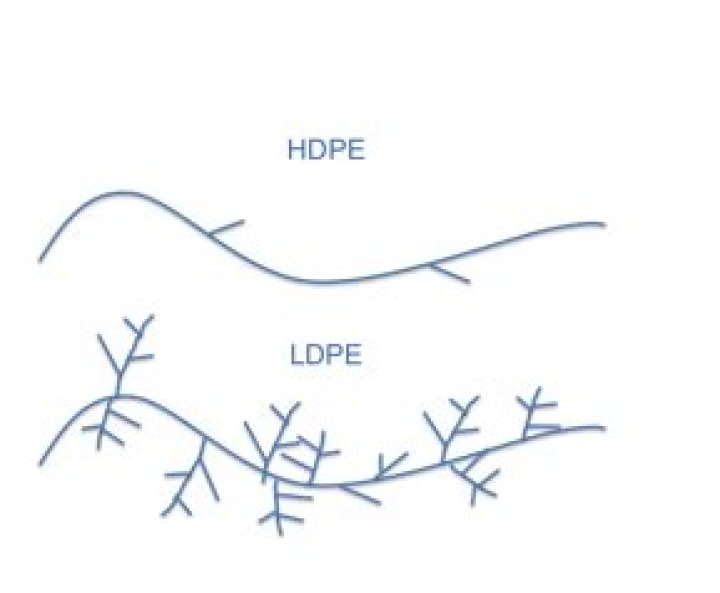
LDPE has a high degree of short and long chain branching, which means that the chains do not pack into the crystal structure as well. It has, therefore, less strong intermolecular forces as the instantaneous-dipole induced-dipole attraction is less.
Does LDPE have a low melting point?
Differences in Physical Characteristics. LDPE is softer and more flexible than HDPE. It also has a lower melting point (115° C) and is more transparent.
Why do plastics have low melting points?
Intermolecular forces are much weaker than the strong covalent bonds in molecules. … The covalent bonds are not broken. Relatively little energy is needed to overcome the intermolecular forces, so simple molecular substances have low melting and boiling points.
Why LDPE is lower melting point than HDPE?
LDPE is a very flexible and tough polyethylene. Compared to HDPE, it has a higher degree of short and long side-chain branching. … However, LDPE has a significantly lower tensile strength, heat deflection temperature and melting point than HDPE.What is melting point of LDPE?
Because the branches prevent the polyethylene molecules from packing closely together in hard, stiff, crystalline arrangements, LDPE is a very flexible material. Its melting point is approximately 110 °C (230 °F).
Why does Polyethene have a range of melting points?
Polymers consist of very long molecules that contain chains of carbon. … There are greater intermolecular forces between the long chains compared with smaller simple molecules. This means that polymers have a higher melting point than many other organic molecules.
Is LDPE a thermoplastic?
Polyethylene (HDPE/LDPE) Polyethylene (PE) is the most widely used thermoplastic polymer for fabricated parts and components. It is available in a variety of grades and formulations to suit different needs.
Why do LDPE and HDPE differ in density?
LDPE’s long- and short-chain branches keep the material from packing tightly in its crystalline form. This gives it less tensile strength than HDPE, but greater ductility. … The molecules are tightly packed together during crystallisation, making HDPE dense and possessing higher resilience than LDPE.Why does Polyethane have a much higher melting point than ethane?
Why does polyethane have a much higher melting point than Ethane? Polyethene long chains have a lot more weak forces of attraction between them. Ethene is a gas at room temperature but polyethene is a solid.
Why is LDPE flexible?The polymer chains of LDPE are highly branched compared to HDPE. This branching prevents the chains from stacking neatly beside each other, reducing the intermolecular forces of attraction. This results in a plastic that is softer and more flexible, but which also has lower tensile strength.
Article first time published onWhy covalent molecules have low melting point?
Covalent molecular They have low melting points and boiling points because the attractions between molecules are easy to overcome. They do not conduct electricity because there are no free charges to move. Some covalent molecular compounds have higher melting points than expected.
Why does water have such a low melting point?
Water molecules are attached to each other by hydogen bonds which is slightly stronger than van dar Waals force; therefore, it is liquid at room temperature. Still it required very low energy to eliminate the H-bond energy compared with the ionic bonds. Therefore, its melting point is comparatively lower.
Why does sucrose have a low melting point?
Thus unlike sodium chloride and most other substances sucrose and lots of other sugars from trioses upwards in numbers of carbons don’t have a boiling point, just a melting point because heat breaks the C−C, C−H, and C−O bonds more in sugars than other organic molecules.
What is the melting point of PLA?
PLA has a glass transition temperature between 50 and 80 °C and a melting temperature between 170 and 180 °C depending on the amount of residual monomer. It is expected to have wide applications because of its excellent mechanical and biodegradable properties, as well as its adjustable hydrolyzability.
What is the melting point of HDPE?
Thermoplastic materials become liquid at their melting point (110-130 degrees Celsius in the case of LDPE and HDPE respectively).
What is LDPE PDF?
Low density polyethylene (LDPE) and High density polyethylene (HDPE) are the two major types of polyethylene which differ in their molecular weight, arrangement, thermal properties and crystalline nature.
Is LDPE thermosetting or thermoplastic?
NamePropertiesPolystyreneLight, hard, stiff, transparent, brittle, with good water resistanceLow density polythene (LDPE)Tough, good resistance to chemicals, flexible, fairly soft, good electrical insulatorHigh density polythene (HDPE)Hard, stiff, able to be sterilised
What are LDPE and HDPE how are they prepared?
LDPE is obtained by the polymerization of ethene under high tension of 1000−2000 atm at 350−570K within the sight of an initiator. We have to know that high-density polyethylene (HDPE) is a thermoplastic polymer created from the monomer ethylene. It is here and there called “polythene” when utilized for HDPE pipes.
How is LDPE manufactured?
LDPE is prepared from gaseous ethylene under very high pressures (up to about 350 megapascals, or 50,000 pounds per square inch) and high temperatures (up to about 350 °C [660 °F]) in the presence of oxide initiators. These processes yield a polymer structure with both long and short branches.
Why does Polyethene have a range of melting points and not a fixed melting point?
Polyethene molecules do not interact strongly with other polyethene molecules. They don’t ‘stick’ together with shared electron density and so any amount of heat will allow molecules to slide right past each other. This underlies the low melting point.
Why do giant covalent substances have high melting points?
Substances with giant covalent structures are solids at room temperature. They have very high melting points and boiling points . This is because large amounts of energy are needed to overcome their strong covalent bonds to make them melt or boil.
Why PP has higher melting point than PE?
Polypropylenes are light in weight. They have a high resistance to cracking, acids, organic solvents and electrolytes. … Monomer of polyethylene is ethylene and monomer of polypropylene is propylene. Polyethylene has a lower melting point compared to the higher melting point of polypropylene.
Why does ethane have higher melting point than methane?
The intermolecular forces at work are van der Waals forces, because these are nonpolar molecules. These van der Waals forces are greater when the molecules are bigger. So, you have that methane has a lower boiling point than ethane, and ethane, a lower one than butane.
Why hexane has higher boiling point than butane?
But, the forces in hexane will be stronger than those in butane because hexane molecules are larger and therefore capable of more extensive Dispersion Forces between each other than the molecules of butane. For this reason, hexane has a higher boiling point than butane.
Why is isobutane lower boiling point than butane?
2. The boiling temperature of isobutane vs butane is about 11°C (19.8°F) lower for isobutane. … Butane has a chain-like structure whilst isobutane has a branched structure. Both have 4 carbon atoms and 10 hydrogen atoms.
Why is LDPE low density?
Properties. LDPE is defined by a density range of 917–930 kg/m3. … The side branches mean that its molecules are less tightly packed and less crystalline, and therefore its density is lower.
Why does LDPE have low density?
LDPE’s polymer chains have side branches. … These side branches stop the polymer molecules from lining up in an orderly fashion. LDPE’s structure is not crystalline. This irregularity is what gives LDPE its lower density.
Why does LDPE branch?
The branching keeps the molecular chains from packing tightly in its crystaline form, so LDPE has less tensile strength but greater ductility. … Less branching means those nicely linear molecules pack together well during crystallization, making HDPE much denser and rigid.
Is LDPE heat resistant?
LDPE is the least expensive, most flexible (more than HDPE), and has good clarity in film form. However, it has low temperature impact resistance and the least heat resistance with a maximum service temperature of 160° to 180°F. LDPE is also susceptible to stress cracking, especially by detergents.
Why is LDPE plastic good?
LDPE (low density polyethylene) is a soft, flexible, lightweight plastic material. LDPE is noted for its low temperature flexibility, toughness, and corrosion resistance.
Is LDPE amorphous?
Amorphous Thermoplastics: These are most of your clear plastics. … These are most of your traditional plastics when you think of plastic “parts”. They include the polyethylene family (LDPE, HDPE, UHMW-PE), Polypropylene, nylon, acetal and fluoropolymers.